Extended Use of Hypothermia in Elderly Patients with Malignant Cerebral Edema as an Alternative to Hemicraniectomy
Article information
Abstract
Background and Purpose
The use of decompressive hemicraniectomy (DHC) for the treatment of malignant cerebral edema can decrease mortality rates. However, this benefit is not sufficient to justify its use in elderly patients. We investigated the effects of therapeutic hypothermia (TH) on safety, feasibility, and functional outcomes in elderly patients with malignant middle cerebral artery (MCA) infarcts.
Methods
Elderly patients 60 years of age and older with infarcts affecting more than two-thirds of the MCA territory were included. Patients who could not receive DHC were treated with TH. Hypothermia was started within 72 hours of symptom onset and was maintained for a minimum of 72 hours with a target temperature of 33°C. Modified Rankin Scale (mRS) scores at 3 months following treatment and complications of TH were used as functional outcomes.
Results
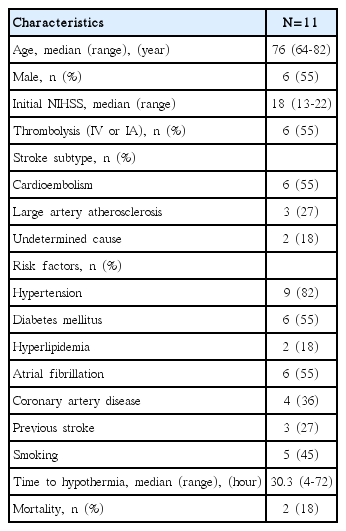
Eleven patients with a median age of 76 years and a median National Institutes of Health Stroke Scale score of 18 were treated with TH. The median time from symptom onset to initiation of TH was 30.3±23.0 hours and TH was maintained for a median of 76.7±57.1 hours. Shivering (100%) and electrolyte imbalance (82%) were frequent complications. Two patients died (18%). The mean mRS score 3 months following treatment was 4.9±0.8.
Conclusions
Our results suggest that extended use of hypothermia is safe and feasible for elderly patients with large hemispheric infarctions. Hypothermia may be considered as a therapeutic alternative to DHC in elderly individuals. Further studies are required to validate our findings.
Introduction
Large hemispheric infarction is a devastating type of stroke accounting for up to 10% of all supratentorial ischemic strokes and has mortality rates of up to 80% [1,2]. The severe mass effect produced by progressive swelling of the infarcted brain tissue and the subsequent inevitable elevation of intracranial pressure typically result in poor neurological outcomes [3]. Conventional medical management, including osmotic therapy, hyperventilation, and barbiturate coma, has not been shown to improve long-term functional outcomes [4-6]. A pooled analysis of three randomized, controlled trials has shown that early decompressive hemicraniectomy (DHC) is beneficial for the treatment of malignant cerebral edema [7-10]. However, approximately 40% of patients with large hemispheric infarctions are older than 60 years [11,12] and the upper age limit in the above analysis was 60.
Recently, a randomized trial demonstrated that DHC improved outcomes and reduced mortality rates in patients older than 60 with large middle cerebral artery (MCA) infarctions [13]. The 6-month mortality rate was lower for patients who underwent hemicraniectomy (33%) than for those who underwent the best possible medical therapy with standardized intensive care (70%). However, DHC was less effective in reducing the mortality rate in older patients than in younger ones (33% vs. 50%).
Older patients with multiple underlying comorbidities may not tolerate general anesthesia or surgical treatment, which presents a major challenge for clinicians in their decision-making process. Moreover, due to cultural difference in attitudes toward surgery in Korea, some patients or caregivers often refuse to receive major surgery, even if it is not contraindicated in their case.
The Cochrane Collaboration Review analyzed eight trials of cooling therapy for the treatment of acute ischemic stroke and reported that therapeutic hypothermia (TH) had no clear benefit in reducing the rates of mortality or functional dependency and was associated with increased risks for cardiac arrhythmias and infections, which have limited its use in clinical practice [14-16]. However, the hypothermic state may activate a number of potentially neuroprotective mechanisms, possibly reducing neurological injury and decreasing cytotoxic edema by preserving cerebrovascular autoregulation [17-19]. The concept of TH has also been suggested to be a promising approach in reducing global cerebral edema and intracranial hypertension due to acute ischemic and hemorrhagic brain injury [5,20].
Hypothermia treatment may be an alternative therapeutic option for patients with large cerebral infarctions who cannot receive DHC. Our study aimed to investigate whether TH can safely reduce or halt the progression of malignant cerebral edema in an elderly population with large hemispheric infarctions.
Methods
Patients
DHC was the first-line therapy for patients with large hemispheric infarcts. However, those patients who declined DHC were then counseled about the use of TH as an alternative treatment option. Written informed consent was obtained from all caregivers prior to patient enrollment into TH treatment. This investigation was approved by the institutional review board.
Patient population
We retrospectively reviewed the clinical and radiographic data from elderly patients aged 60 years or older who were diagnosed with a large acute hemispheric infarction and were treated with TH between February 2011 and August 2012. Seoul National University Bundang Hospital’s stroke data registry was used for analysis. Patients were enrolled if they met all of the following criteria: age ≥60 years, with acute ischemic stroke, hospital admission within 12 hours of stroke symptom onset, and initial National Institutes of Health Stroke Scale (NIHSS) score >10. Specific neuroimaging criteria included the following: large hemispheric infarction involving more than two-thirds of the MCA territory, involvement of the basal ganglia on diffusion-weighted magnetic resonance imaging scans within 12 hours of symptom onset, and parenchymal hypodensity involving more than two-thirds of the MCA territory with a midline shift evident on computed tomography (CT) scans within 72 hours of onset.
Patients were ineligible if they demonstrated any of the following exclusion criteria: severe cardiac dysarrhythmia or a corrected QT (QTc) interval >450 ms, platelet count <75,000/mm3, known coagulopathy (prothrombin time international normalized ratio >1.3) or active bleeding, severe sepsis and septic shock, preexisting illness with a life expectancy of <6 months, previous modified Rankin Scale (mRS) score >4, concurrent monoamine oxidase inhibitor usage, or pregnancy [21].
Data collection
We reviewed clinical data, including patient demographics, diagnosis, Trial of ORG 10172 in Acute Stroke Treatment classification, vascular risk factors (hypertension, diabetes mellitus, hyperlipidemia, smoking, previous stroke, and previous cardiac disorders including arrhythmia), stroke severity as measured by NIHSS score at baseline and discharge, and long-term functional outcome 3 months following treatment as measured by the mRS.
Radiologic data included an initial magnetic resonance imaging scan on admission and follow-up computed tomography (CT) scans before and after TH. The follow-up CT scans were used to detect any hemorrhagic transformation and to quantify the magnitude of the midline shift 1 day and 3 days after completing TH. Hemorrhagic transformation was categorized into four different subtypes (hemorrhagic infarction 1 or 2, and parenchymal hemorrhage 1 or 2) according to the European Cooperative Acute Stroke Study I and II criteria [22,23]. The progression of cerebral edema was quantified by measuring lateral displacement of the calcified pineal gland from the midline on CT scans [24].
Physiological data included heart rate, blood pressure, and body temperature, which were measured every hour. Timeline data consisted of admission and discharge date, stroke onset, time of symptom onset to TH initiation, and time to target temperature. Laboratory data included white blood cell count, hematocrit and hemoglobin levels, platelet count, electrolytes (sodium, potassium, chloride, and phosphate), glucose, creatinine, albumin, creatinine kinase, aspartate transaminase, alanine transaminase, prothrombin time, activated partial thromboplastin time, and arterial blood gas. Urinalysis, electrocardiography, and chest radiography were also performed. Daily chest radiographs and microbiological pancultures (urine, blood, and sputum) were reviewed for infection status. Adverse events were adapted from the National Acute Brain Injury Study [25], which included cardiovascular complications, infections, electrolyte imbalances, and bleeding complications or coagulopathies, pulmonary edema, and liver function abnormalities.
Clinical assessment
Neurological deficits on admission and discharge were assessed using the NIHSS score. The functional status of each patient was assessed 3 months after treatment using mRS scoring. All clinical assessments were recorded by cerebrovascular neurologists. Complications arising from TH were recorded to determine safety and feasibility outcomes.
Hypothermia treatment
All patients were admitted to the neurological intensive care unit and treated according to the standardized institutional hypothermia protocol after obtaining informed consent. Patients were cooled with an external cooling device using cold water-circulating wrapping garments (Arctic Sun Temperature Management System; Medivance, Louisville, CO, USA) and subsequent use of 500 mL to 1 L of cold (4.0°C) saline infused intravenously within 30 to 60 minutes after enrollment. Core temperature was measured with an esophageal probe. After the core temperature reached the target temperature of 33°C, the Arctic Sun system automatically maintained the target temperature by regulating the temperature of the circulating water. Core temperature was continuously monitored and was recorded every hour. Invasive monitoring equipment, such as an arterial line or a central venous line, was used for those who needed close hemodynamic monitoring.
All patients treated with TH were intubated and adequately sedated. We adopted the Columbia University Anti-shivering Protocol [26], which included both universal and patient-specific therapies. Initially, acetaminophen (650 mg every 4 hours) and buspirone (30 mg every 8 hours) were used as anti-shivering agents, and a skin counter-warming system (Bear Hugger, Arizant Healthcare Inc.; Eden Prairie, MN, USA) was applied. If shivering developed, meperidine (50-100 mg bolus followed by 12.5-50 mg/h) or propofol infusions (75-300 mg/h) were used. If shivering persisted, a dexmedetomidine infusion (loading dose 1 µg/kg over 10 minutes, followed by an infusion of 0.3-1.5 µg/kg/h) was used, and finally, a continuous infusion of vecuronium (0.1 mg/kg/h) was administered for refractory shivering cases.
All patients underwent follow-up CT scans before and after TH to visually assess the degree of cerebral edema. After obtaining stable clinical and CT findings for 24 hours, a passive and slow rewarming process was initiated. The rate of rewarming was set at 0.1°C/h.
Results
Patient characteristics
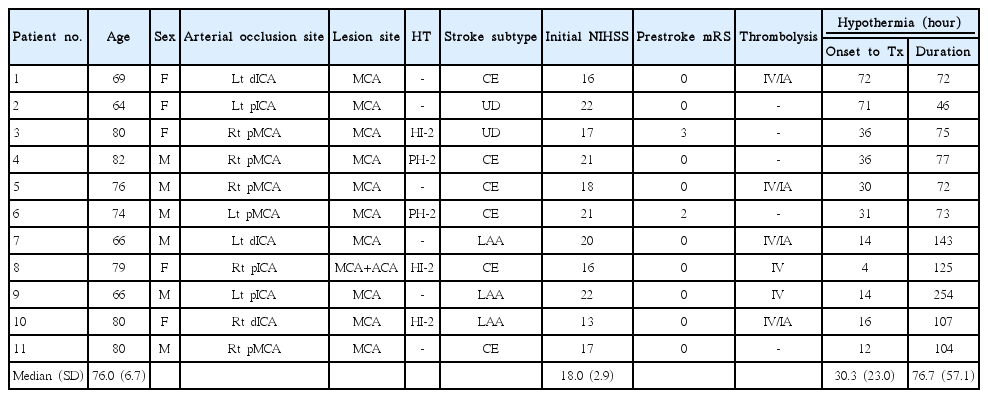
A total of 199 patients with MCA infarctions between February 2011 and August 2012 were screened. Thirty-four patients had malignant MCA infarctions. Five patients (15%) underwent DHC, 13 (38%) received conservative treatment, 16 (47%) were treated with TH, and 11 (32%) were eligible for our study. Baseline patient characteristics are summarized in Table 1. The median age was 76 (range, 64-82 years) and the median NIHSS score was 18 (range, 13-22) upon admission. Six (55%) of the 11 patients underwent intravenous or intra-arterial thrombolysis. Based on Trial of ORG 10,172 in Acute Stroke Treatment subtype classification, 6 (55%) of 11 patients had a cardioembolism, 3 (27%) had large artery atherosclerosis, and 2 (18%) were of undetermined etiology. One patient had two potential causes of stroke (large artery atherosclerosis and cardioembolism), and the other patient was not fully evaluated due to death (undetermined etiology, negative evaluation). Nine (82%) of 11 patients exhibited hypertension and 3 (27%) had previously experienced stroke. Premorbid mRS scores were 0 for 9 patients, 2 for 1 patient, and 3 for 1 patient. Hemorrhagic transformation was detected in 5 (45%) of 11 patients before the initiation of hypothermia, and 4 (80%) out of these 5 patients experienced an additional mass effect due to hemorrhagic transformation (parenchymal hemorrhage 1 or 2).
Feasibility of therapeutic hypothermia
TH was initiated at a median of 30.3±23.0 hours after stroke onset (Table 2). The median time to reach core target temperature (33°C) after cold saline infusion was 4.0±2.4 hours. TH was maintained for a median of 76.7±57.1 hours (range, 46-254 hours).
Outcomes
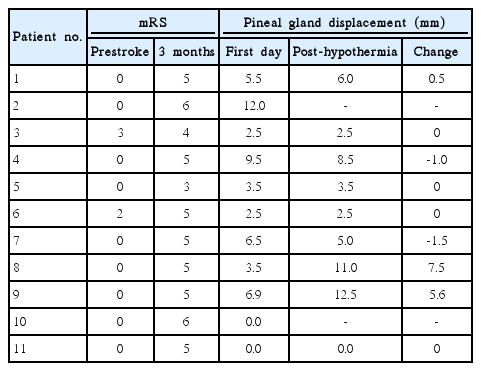
The mean mRS score 3 months after treatment was 4.9±0.8 for all patients and 4.7±0.7 for survivors (Table 3). There were two deaths (18%) due to either progression of cerebral edema (case 2) or sudden cardiac arrhythmia after rewarming (case 10). One (9%) of the 11 patients treated with TH achieved a favorable functional outcome (mRS 3).
All patients who exhibited pineal gland displacement >5 mm on the first day of TH experienced poor outcomes (mRS 5 or 6) and the degree of the lateral displacement of the pineal gland was more significant in those who died (case 2: 12-mm pineal gland displacement). Two (33%) of 6 patients with <5 mm of pineal gland displacement exhibited an mRS of 3 or 4 three months after treatment, suggesting that the magnitude of pineal gland displacement on the first day of TH corresponded with mortality and functional outcome.
Safety and complications
Shivering was observed in all TH patients (Table 4). Bradycardia was frequently noted during the cooling phase (45%, n=5), but was transient in nature and asymptomatic in all cases. Five patients (45%) who had preexisting atrial fibrillation developed further cardiac arrhythmias during hypothermia, 2 (18%) developed a non-sustained ventricular tachycardia, and 3 (27%) experienced atrial fibrillation with rapid ventricular responses. No patient required urgent anti-arrhythmic therapy. Hypotension was noted in 2 (18%) cases and was thought to be due to hypothermia-induced diuresis and relative intravascular volume depletion. Six (55%) patients developed pneumonia, but responded favorably to appropriate antibiotic therapies.
Electrolyte imbalances were detected in most patients during TH. Serum sodium was altered in 55% of patients (hypernatremia, n=5; hyponatremia, n=1). Hypokalemia was observed in 82% of patients (n=9) and 1 patient developed rebound hyperkalemia during the rewarming phase. Hypophosphatemia was also a common side effect (82%, n=9). Minor bleeding complications, such as hemoptysis (n=2) and hematuria (n=2), were observed, but were easily controlled. No major coagulopathy was induced by TH.
Discussion
Our results suggest that TH is well-tolerated and feasible for clinical use for extended periods (i.e., >72 hours) in patients older than 60 years of age with large acute hemispheric infarctions.
In a pooled analysis of three European trials of hemicraniectomy (DESTINY, DECIMAL, and HAMLET) involving younger patients (age <60 years), mortality rates for conservative treatment and surgery were 77% and 22%, respectively, at 12 months after treatment [7]. In patients older than 60 years of age (median, 70 years; range, 61-82 years), hemicraniectomy reduced 6-month mortality by 33% and 1-year mortality by 43%, as compared to 70% and 76% in a control group [13]. Our overall 18% mortality rate in elderly patients treated with TH is comparable to the results of a previous pooled analysis of DHC (22%) in young patients, but slightly higher than that of a study by Georgiadis et al. [27], who reported a 12% mortality rate in 17 patients who received DHC. The above randomized controlled trials were conducted with relatively young patients (<60 years old in most cases) and the mortality rate of patients who underwent DHC was notably high at approximately 20-25% [8-10]. Although our study is not appropriately powered, our results indicate that TH may be a safe and feasible alternative to DHC in elderly patients with malignant hemispheric edema, especially given the inevitable risks and potential complications associated with surgery.
The feasibility and safety of hypothermia were previously demonstrated in the literature [15,16]. The ICTuS trial showed that hypothermia using an endovascular method decreased acute post-ischemic cerebral edema [20]. The authors of that trial demonstrated the feasibility and safety of combining endovascular hypothermia with intravenous thrombolysis [28]. Although the duration of hypothermia was limited to 72 hours in the previous studies, we opted to continue cooling for much longer durations (up to 254 hours) in a larger number of patients. One patient (case 9) initially underwent TH for 155 hours with clinical success in halting malignant edema. Rebound intracranial pressure crises and edema were not infrequent and were observed during the passive rewarming phase. As a result, TH was restarted for an additional 99 hours for the patient who developed a recurrence of cerebral edema, as indicated by clinical and radiographic data.
As observed in previous studies, our results demonstrate that TH is associated with a number of specific complications, such as pneumonia, bradycardia, arrhythmia, and hypokalemia, which require intensive monitoring and management in the neurological intensive care unit. In our study, the rate of complications due to TH increased as the duration of hypothermia increased, although all complications were well-tolerated. Six of 11 patients developed pneumonia and an increased rate of pneumonia was observed as the duration of hypothermia increased.
Our study provides a number of useful findings. First, a longer duration of TH (more than 72 hours) appeared to be feasible in these patients. Moderate hypothermia was induced effectively and maintained for more than 72 hours in all patients. Second, the timing of TH may be important. It is likely that the patient in case 2 died due to a delayed implementation of therapy. We also observed a relationship between the magnitude of pineal gland displacement (on the first day of stroke) and functional outcome: no patients with a pineal gland displacement >10 mm experienced a positive outcome. However, it remains unknown whether a positive outcome could have been observed if TH was initiated prior to the onset of significant edema and/or severe pineal gland displacement.
Our study has a number of limitations. The predominant limitations are that this is a single-center, retrospective, observational study with a small sample size. A larger, multicenter, prospective study with more extensive data that exclude all potential confounders is warranted to further investigate the utility of TH in elderly individuals.
Conclusions
Our study provides data that are useful for the clinical decision-making process. When considering surgery for an elderly patient with a malignant MCA infarction, hypothermia may potentially be a reasonable alternative, particularly for those who may not be able to tolerate the risks of surgery. Based on our results and the available literature, a larger randomized controlled trial of hypothermia is required to validate our findings in an elderly population.
Notes
The authors have no financial conflicts of interest.