Effects of Triflusal and Clopidogrel on the Secondary Prevention of Stroke Based on Cytochrome P450 2C19 Genotyping
Article information
Abstract
Background and Purpose
To compare the efficacy and safety of antiplatelet agents for the secondary prevention of ischemic stroke based on cytochrome P450 2C19 (CYP2C19) polymorphisms.
Methods
This study was a prospective, multicenter, randomized, parallel-group, open-label, blind genotype trial. First time non-cardiogenic ischemic stroke patients were enrolled and screened within 30 days. Participants were randomized to receive either triflusal or clopidogrel for secondary stroke prevention. The primary outcome was the time from randomization to first recurrent ischemic stroke or hemorrhagic stroke.
Results
The required sample size was 1,080 but only 784 (73%) participants were recruited. In patients with a poor CYP2C19 genotype for clopidogrel metabolism (n=484), the risk of recurrent stroke among those who received triflusal treatment was 2.9% per year, which was not significantly different from those who received clopidogrel treatment (2.2% per year; hazard ratio [HR], 1.23; 95% confidence interval [CI], 0.60–2.53). In the clopidogrel treatment group (n=393), 38% had good genotypes and 62% poor genotypes for clopidogrel metabolism. The risk of recurrent stroke in patients with a good CYP2C19 genotype was 1.6% per year, which was not significantly different from those with a poor genotype (2.2% per year; HR, 0.69; 95% CI, 0.26–1.79).
Conclusions
Whilst there were no significant differences between the treatment groups in the rates of stroke recurrence, major vascular events, or coronary revascularization, the efficacy of antiplatelet agents for the secondary prevention of stroke according to CYP2C19 genotype status remains unclear.
Introduction
In the absence of an effective secondary prevention strategy, patients with ischemic stroke (IS) have a high probability (4–16% per year) of experiencing a second atherosclerotic vascular event [1]. Oral antiplatelet therapy is a widely used secondary prevention strategy for non-cardiogenic IS [2,3]. However, several studies have shown that platelet inhibition by clopidogrel exhibits patient-to-patient variability [4]. Factors associated with clopidogrel response variability include clinical and genetic variables, as well as drug interactions. The hepatic cytochrome P450 (CYP) enzymatic system metabolizes approximately 80% of all clinical drugs, and is required for the bioactivation of clopidogrel; CYP2C19 generates an active oxidized metabolite of clopidogrel that exerts antiplatelet activity by inhibiting P2Y12 receptors [5]. Though numerous studies have demonstrated an association between clopidogrel resistance and adverse ischemic events, this phenomenon has yet to be confirmed in a large-scale trial.
For the secondary prevention of IS, triflusal has a comparable antiplatelet effect to aspirin and offers a lower risk of bleeding [6,7]. Triflusal does not require oxidation by the hepatic CYP system for the generation of active metabolites; rather, triflusal is deacetylated during hepatic first-pass metabolism to form 2-OH-4-trifluoromethyl benzoic acid. This metabolite irreversibly inhibits cyclooxygenase-1 (COX-1) and reduces the production of thromboxane B2 (TXB2) [2].
No clinical study to date has evaluated the relationship between stroke recurrence during secondary prevention of IS and CYP2C19 genotype status. Accordingly, the Comparison of Triflusal and Clopidogrel Effects in Secondary Prevention of stroke based on cytochrome P450 2C19 genotyping (MAESTRO) study was designed to compare the efficacy and safety of antiplatelet agents for the secondary prevention of IS based on CYP2C19 polymorphisms.
Methods
Study design and participants
The MAESTRO study methods have been published previously [8]. This study was a prospective, multicenter, randomized, parallel-group, open-label, blind genotype trial conducted at 18 tertiary-care hospitals in South Korea, between March 15, 2010 and December 31, 2014. Participants were eligible if they were 20 years of age or older and had their first time non-cardiogenic IS within 30 days prior to screening. Non-cardiogenic IS was classified according to the Trial of ORG 10172 in the Acute Stroke Treatment (TOAST) classification system. The main exclusion criteria were: contraindications to trial drugs, a high-risk potential of cardiac source of embolism, any non-atherosclerotic vasculopathies, use of anticoagulants or two or more antiplatelet agents, congestive heart failure, impaired renal or hepatic function, anemia or thrombocytopenia, bleeding diathesis or cancer, and pregnancy or lactation [8]. The study protocol was approved by the institutional review board of each participating hospital. All patients provided written informed consent prior to study participation.
Randomization and masking
Eligible participants were randomized in a 1:1 ratio to either the triflusal group or clopidogrel group. Randomization was stratified by clinical site via random permuted blocks in sizes of four or six. Each screen-eligible participant was randomly assigned through a secure web-based registration system with telephone back-up. The study had an open-label design; both the investigator and patient were aware of the study drug, but genotype data were masked until the end of the trial.
Procedures
During the trial, participants received either 300 mg triflusal twice per day (600 mg/day) or 75 mg clopidogrel once daily. Initial loading of the study drug, proton pump inhibitor, and use of a concomitant antiplatelet or anticoagulant drug were strictly prohibited. All patients received appropriate medical treatment at the discretion of the attending neurologist, including anti-hypertensive and/or statin treatment, and rigorous control of other vascular risk factors [8].
CYP2C19 genotype status was assessed using the Seeplex CYP2C19 ACE genotyping system (Seegene, Seoul, Korea) and Real-Q CYP2C19 genotyping kit (Biosewoom, Seoul, Korea). The major alleles of the CYP2C19 gene are *1, *2, *3, and *17, and about 55% of Asians and 30% of Caucasians have a loss-offunction mutation in one or more alleles (*2 or *3) [9]. The Seeplex CYP2C19 ACE Genotyping system identified two single nucleotide polymorphisms (*2, *3 alleles) and the Real-Q CYP2C19 genotyping kit detected the *17 polymorphism. Each participant was classified as an ultrarapid metabolizer (UM: *1/*17, *17/*17), extensive metabolizer (EM: *1/*1), intermediate/unknown metabolizer (*1/*2, *1/*3 and *2/*17, *3/*17), or poor metabolizer (PM: *2/*2, *2/*3, *3/*3) based on their CYP2C19 genotype status. UM or EM status patients were allocated into the good genotype group for clopidogrel metabolism, and intermediate/unknown metabolizer, or PM status patients, were allocated into the poor genotype group.
Outcome measures
The primary outcome was the time from randomization to first recurrent IS or hemorrhagic stroke. Evidence of stroke included: a) symptoms lasting more than 24 hours, or b) symptoms completely resolved within 24 hours based on neuroimaging in the clinically relevant brain area. There were three pre-specified secondary outcomes: 1) time to first IS, 2) time to first myocardial infarction (MI) or coronary revascularization, and 3) time to first major vascular event (i.e., stroke, MI, or vascular death). All suspected outcomes were adjudicated by the Central Independent Adjudication Committee, based on anonymized written descriptions of the outcome event and ancillary investigations. The committee was masked to treatment allocation. Safety was evaluated by regular examination and laboratory testing after enrolment throughout the study. An independent data safety monitoring committee evaluated adverse drug events, and reviewed the safety of the study.
Statistical analysis
Direct comparison studies were not available to support a comparison of triflusal and clopidogrel monotherapy. Based on the TACIP study, an annual stroke recurrence rate of 3.85% was expected in the triflusal treatment group [10]. According to the PRoFESS trial, an annual stroke recurrence rate of 3.52% was expected in the clopidogrel treatment group [11]. Among clopidogrel-treated patients in the TRITON-TIMI 38 study, increased risk of stroke was observed in patients carrying a CYP2C19 reduced-function allele compared with non-carriers (0.88% vs. 0.24%; hazard ratio [HR], 3.93; 95% confidence interval [CI], 0.66–23.51) [12]. Based on these results, this study assumed an annual stroke recurrence rate of 7.74% in the clopidogrel group and 3.85% in the triflusal group in patients allocated to the poor genotype group for clopidogrel metabolism. Thus, at least 594 participants with a poor genotype were required for the primary analysis based on a log-rank test to detect a 50% risk reduction in the triflusal group with 80% power, a two-sided type I error of 5.0%, a recruitment period of two years, a minimum follow-up period of 24 months, and a discontinuation rate of 20%. Based on a report indicating that 55% of Korean participants belong to the poor genotype group [13], the required sample size was 1,080.
Efficacy analyses were performed on both modified intention-to-treat (mITT) and the per-protocol (PP) populations. The mITT population included patients who had taken at least one dose of the study drug after randomization. The PP population included patients who completed the study schedule without a protocol violation and had >80% medication adherence. Safety analyses were performed on the mITT population. Cumulative incidences of primary and secondary outcomes were estimated by the Kaplan-Meier method, and differences between groups were assessed using log-rank tests. Cox proportional-hazards models were used to calculate hazard ratios. Subgroup analyses were performed to test whether CYP2C19 polymorphisms were associated with the primary outcome in the treatment group. Statistical analyses were performed with SAS version 9.4 (SAS Institute, Cary, NC, USA).
Results
Patient characteristics
The required study sample size was 1,080; however, only 784 participants (73%) were recruited during the trial, despite a one-year study period extension to accommodate slow enrolment. Between March 2010 and December 2012, a total of 2,875 patients were assessed; 795 participants met the inclusion criteria and underwent randomization. Of these, 784 (99%) received at least one dose of the trial drug and were included in the mITT population, and 597 (76%) completed the trial and were included in the PP population (Figure 1). The median duration of follow-up was 2.7 years (range, 0–4.7). The numbers of withdrawals were similar between the two groups. Table 1 summarizes the baseline characteristics of enrolled patients. The mean age was 61 years, and 32% of participants were women. Sixty-one percent of participants had a history of hypertension, 29% had diabetes, 28% had dyslipidemia, and 40% were current tobacco smokers. The median time from qualifying stroke to randomization was 8 days, and 76% of patients were randomized within 14 days. There were no significant differences in the times from qualifying stroke to randomization between the treatment groups. Brain MRI and diffusion-weighted imaging were performed in 98% of patients. All patients completed at least one vascular imaging study (86% magnetic resonance angiography, 28% computed tomography angiography, and 4% cerebral angiography). The most frequent stroke subtype was lacunar infarction, which occurred in 52% of the population, followed by stroke of undetermined etiology in 27% of the population, and large artery atherosclerosis in 21% of the population (Table 1). During follow-up, 86% of patients were taking a statin and 69% were taking an anti-hypertensive drug.
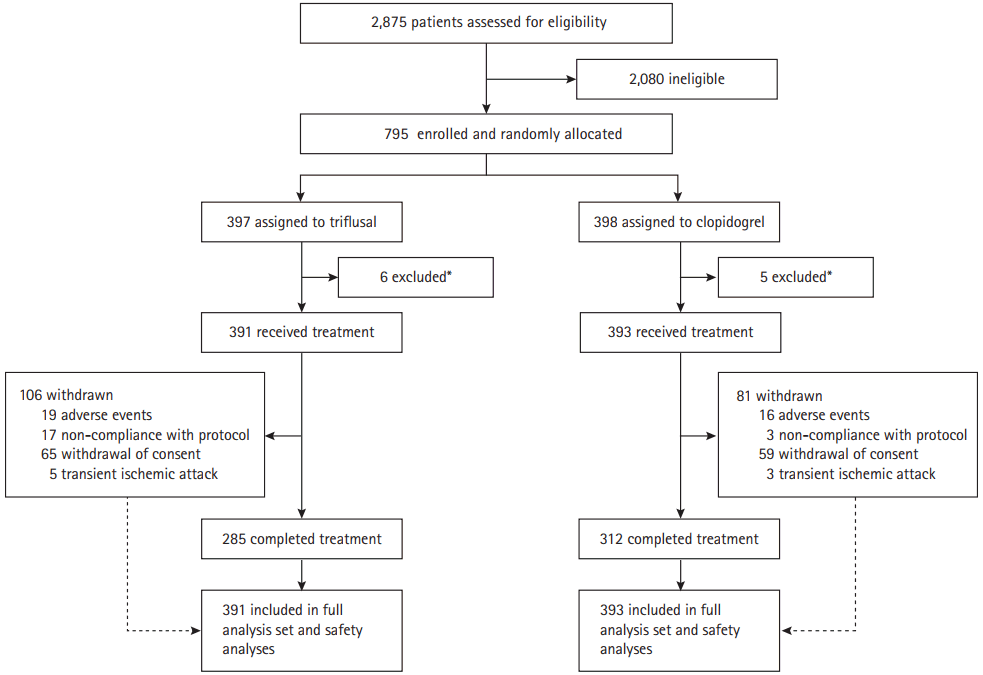
Design of the MAESTRO study. MAESTRO, stroke based on cytochrome P450 2C19 genotyping. *Withdrawal of consent before genotyping.
Clinical outcomes
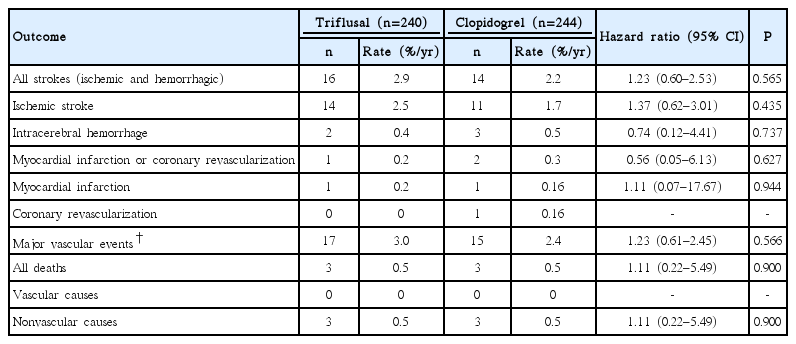
Within the poor genotype group for clopidogrel metabolism (484 participants), there were no statistically significant differences in baseline characteristics according to treatment group. Thirty (6.2%) had recurrent stroke: 25 (83%) experienced an IS and 5 (17%) experienced an intracerebral hemorrhage (ICH). The risk of recurrent stroke among patients assigned to the triflusal treatment group was 2.9% per year, and was not significantly different from that among patients assigned to the clopidogrel treatment group (2.2% per year; HR, 1.23; 95% CI, 0.60–2.53; Table 2 and Figure 2). There was no significant difference in the reduction of recurrent IS risk according to treatment group (HR, 1.37; 95% CI, 0.62–3.01). In the triflusal treatment group, there was no significant reduction in the risk of a major vascular event (HR, 1.23; 95% CI, 0.61–2.45). Moreover, no difference in all-cause mortality was observed between treatment groups (HR, 1.11; 95% CI, 0.22–5.49). Finally, there was no heterogeneity of treatment effect on the primary outcome, according to age, sex, smoking, stroke subtype, or between the pre-specified subgroups (P>0.05 for all comparisons; Supplementary Figure 1).
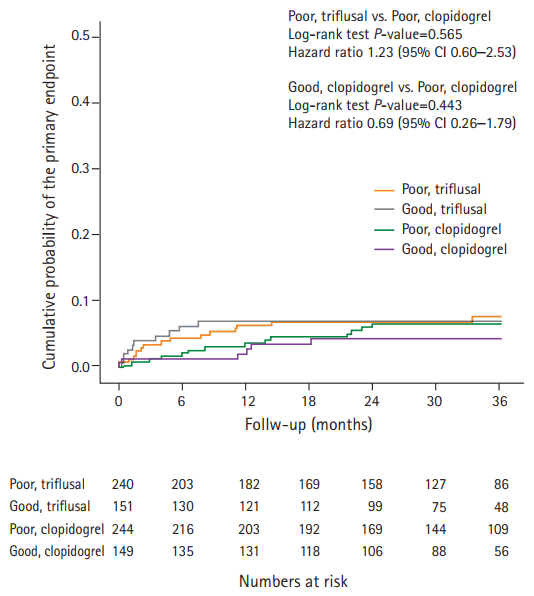
Differences in the primary endpoint according to CYP2C19 geno-type and treatment. CYP2C19, cytochrome P450 2C19; CI, confidence interval.
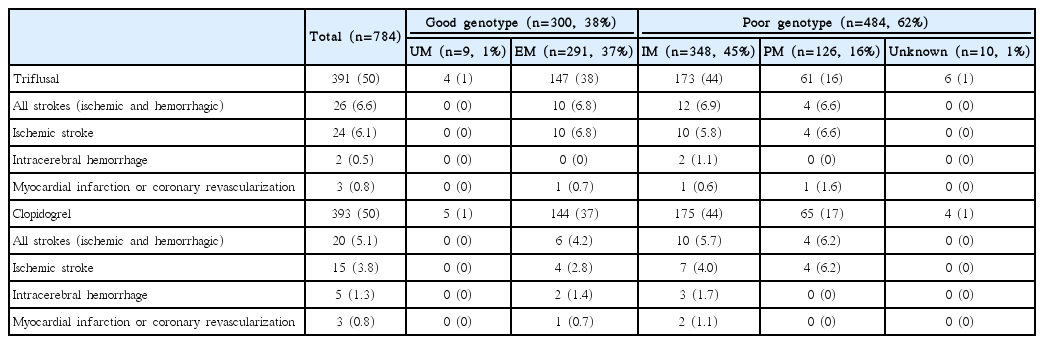
Of 393 participants in the clopidogrel treatment group, 149 (38%) were assigned to the good genotype group and 244 (62%) were assigned to the poor genotype group for clopidogrel metabolism. There were no statistically significant differences in baseline characteristics according to genotype group (Table 1). A total of 20 (5.1%) patients had a recurrent stroke: 15 (75%) experienced an IS and 5 (25%) experienced an ICH. The risk of recurrent stroke among patients in the good genotype group was 1.6% per year and was not significantly different from that among patients in the poor genotype group (2.2% per year; HR, 0.69; 95% CI, 0.26–1.79) (Table 3 and Figure 2). In the good genotype group, there was a non-significant 41% decrease in the relative risk of recurrent IS (HR, 0.59, 95% CI, 0.19–1.85), and no significant reduction in the risk of a major vascular event (HR, 0.64; 95% CI, 0.25–1.66). Subgroup analyses showed that there was a significant treatment-by-subgroup interaction for sex in patients randomized to the clopidogrel group based on genotype (P=0.027). There was a trend towards a reduction in risk for the primary outcome in men in the good genotype group, whilst women in the good genotype group showed an increased risk for the primary outcome (Supplementary Figure 2).
The most frequent genotype in the entire study population was intermediate metabolizer (348, 45%), followed by EM (291, 37%), PM (126, 16%), unknown (10, 1%), and UM (9, 1%). These results are consistent with previous findings [13]. There were also no significant differences in stroke recurrence, major vascular events, or coronary revascularization regarding treatment and genotype (Table 4).
The PP analysis, which included 285 patients from the triflusal treatment group and 312 patients from the clopidogrel treatment group, showed results consistent with those above. Of 386 participants in the poor genotype group, 8.2% had a recurrent stroke. The risk of recurrent stroke among patients assigned to the triflusal treatment group was 3.2% per year, and was not significantly different from that among patients assigned to the clopidogrel treatment group (2.3% per year; HR, 1.33; 95% CI, 0.65–2.71). In the clopidogrel treatment group, 117 participants (38%) were assigned to the good genotype group and 195 (62%) were assigned to the poor genotype group for clopidogrel metabolism. Of participants in the clopidogrel treatment group, 20 (6.4%) had a recurrent stroke. The risk of recurrent stroke among patients in the good genotype group was 1.7% per year, and was not significantly different from that among patients in the poor genotype group (2.3% per year; HR, 0.71; 95% CI, 0.27–1.86).
Safety
During follow-up, there were no significant differences in the hemorrhage rates between the treatment groups (Table 5). In the poor genotype group, the rates of all types of hemorrhages were 2.1% per year in the triflusal treatment group, and 2.2% per year in the clopidogrel treatment group (HR, 0.97; 95% CI, 0.48–2.24). In the clopidogrel treatment group, the rates of all types of hemorrhages were 3.3% per year in the good geno-type group and 2.2% per year in the poor genotype group (HR, 1.66; 95% CI, 0.78–3.55).
Discussion
Despite an extended enrolment period, this trial failed to meet its recruitment goal due to slow enrolment, and was therefore discontinued. The main reason for this may have been that many investigators did not enroll patients with severe atherosclerotic changes, but instead elected to implement early aggressive dual antiplatelet therapy. Therefore, about half of the enrolled patients had lacunar infarction. Prior to the MAESTRO trial, no direct comparison study had addressed the relationship between CYP2C19 polymorphisms and stroke recurrence during secondary prevention of IS [14]. Since the initiation of the MAESTRO trial, the SPS3 trial subgroup analysis identified significant differences in stroke recurrence according to CYP2C19 genotype-inferred metabolizer status in white subcortical stroke patients receiving aspirin and clopidogrel [15]. Notably, no significant differences were observed when patients from all race groups were combined. The recent CHANCE trial substudy demonstrated that the use of clopidogrel plus aspirin, compared with aspirin alone, reduced the risk of first-time stroke in a subgroup of patients who were not carriers of a CYP2C19 loss-of-function allele [16]. In this trial, stroke occurred in 8.2% of patients in the clopidogrel-aspirin group and 11.7% of those in the aspirin group. However, this study was performed exclusively among Chinese patients with minor IS or transient ischemic attack [17]. Only 42% of patients were receiving lipid-lowering agents, 35% were receiving anti-hypertensive drugs, and 25% were receiving a form of traditional Chinese medicine [18]. During the SPS3 trial, the risk of recurrent stroke was 2.7% per year among participants taking aspirin and 2.5% per year among those taking aspirin and clopidogrel dual therapy; however, 85% of patients in this trial were also using a statin [19].
In the present study, we observed a 41% decrease in the relative risk of recurrent IS in the good CYP2C19 genotype group for clopidogrel metabolism, yet with a wide 95% CI in the clopidogrel treatment group. Similarly, several clinical trials in coronary artery disease have failed to demonstrate associations between CYP2C19 polymorphisms and clinical outcomes [20,21]. In the TRILOGY ACS trial, CYP2C19 functional variants were not successfully associated with the composite outcome of cardiovascular death, MI, or stroke [22]. The results of our study may be inconclusive but, in an ad hoc manner, we calculated a conditional power (CP), or the chance of obtaining a statistically significant result at the end of the trial given the data so far. Even if the MEASTRO study was not prematurely stopped, the CP for obtaining a significant effect for CYP2C19 genotype was as low as 4.2%. Thus, it can be hypothesized that the findings observed in the CHANCE trial are not easily replicated in populations with a higher adherence to risk factor control.
We found an unexpected interaction between sex and clopidogrel treatment among patients in the good genotype group. Men receiving clopidogrel treatment in the good genotype group showed a trend toward reduced risk for the primary outcome. Ex vivo studies have demonstrated that women are more often hyporesponsive to clopidogrel than men; however, the clinical implications and validity of this observation remain uncertain [23]. A recent meta-analysis showed evidence of similar gender-related benefits (major cardiovascular events, MI, or stroke) and risks (major bleeding) in vascular disease; the random-effect estimates ratio of relative risks (rRR) across studies was not statistically different according to male sex (rRR, 0.97; 95% CI, 0.90–1.04) [24]. Further multinational pragmatic trials may be essential for exploring alternative predictors of response to P2Y12 receptor inhibitor therapy with respect to baseline risk factors, ethnicity, or prior disease.
There were several limitations to this study, particularly sample size calculation and subsequent recruitment. We could not refer to a previous clinical trial on stroke recurrence according to CYP2C19 genotype status during clopidogrel therapy. We used the results from the TRITON-TIMI 38 and PRoFESS studies to calculate annual stroke recurrence, but a low stroke rate in TRITON-TIMI 38 study resulted in a large HR and wide CI for stroke in the poor genotype group (HR, 3.93; 95% CI, 0.66–23.51). Second, the actual annual rate of stroke recurrence observed in our study was less than one third of the anticipated value according to our study design. A low stroke rate may have been related to the use of statins and anti-hypertensive drugs by the majority of included participants, which was similar to the rates in other recent studies for the prevention of recurrent stroke [19]. Third, the effect of antiplatelet agents may vary according to ischemic stroke subtype; in this trial, 52% of participants had lacunar infarction, which may have been of pure atherothrombotic origin [25]. Finally, this trial enrolled only Korean patients, limiting the generalizability of our findings to other geographic regions. These limitations should be considered during the interpretation of our data.
Conclusions
There were no differences in stroke recurrence, major vascular events, or coronary revascularization between triflusal and clopidogrel therapy in patients with a poor genotype for clopidogrel metabolism. While we observed a non-significant 41% decrease in relative risk of recurrent IS associated with a good clopidogrel metabolism genotype, our study was not sufficiently powered to detect a statistically significant difference. The results of this study were inconclusive in terms of antiplatelet agent efficacy for the secondary prevention of stroke according to CYP2C19 genotype status. Genes have been found to alter patient responses to pharmacological agents and influence clinical disease outcomes but, currently, there are no standardized treatment recommendations for patients with CYP2C19 genotypes associated with poor clopidogrel metabolism [26,27]. Our findings support the need for additional efficacy and safety data to enable physicians to make individual treatment decisions for the secondary prevention of stroke.
Notes
The authors have no financial conflicts of interest.
Acknowledgements
This study was funded by MyungIn Pharmaceuticals, which had no role in the trial design, data collection, analysis, interpretation, or writing of the report. The corresponding author reviewed the trial report, had full access to all study data, and took final responsibility for the decision to submit the work for publication. All other authors were investigators who had full access to the data.
Supplementary Material
Supplementary materials related to this article can be found online at https://doi.org/10.5853/jos.2017.01249.
Hazard ratios for the primary outcome in subgroups of participants with a poor genotype for clopidogrel metabolism. CI, confidence interval; LAA, large artery atherosclerosis; SUD, stroke of undetermined aetiology.
Hazard ratios for the primary outcome in subgroups of participants who received clopidogrel treatment. CI, confidence interval; TOAST, Trial of ORG 10172 in the Acute Stroke Treatment; LAA, large artery atherosclerosis; SUD, stroke of undetermined aetiology; N/A, not available.