Recent Advances in Primary and Secondary Prevention of Atherosclerotic Stroke
Article information
Abstract
Atherosclerosis is a major cause of ischemic stroke that can be effectively prevented with appropriate lifestyle modifications and control of cardiovascular risk factors. Medical advances in recent years along with aggressive cardiovascular risk factor modifications have resulted in decreased recurrence rates of atherosclerotic stroke. Non-statin lipid-lowering molecules have recently shown clinical benefit and are recommended for very high-risk patients to reduce their risk of stroke. Aggressive hypertension treatment is crucial to reduce atherosclerotic stroke risk. Advances in antithrombotic treatments include combinations of antiplatelets and new antiplatelet agents in the acute phase post-stroke, which carries a high risk of recurrence. Intensive medical treatment has also limited the indications for carotid interventions, especially for asymptomatic disease. Intracranial atherosclerotic disease may provoke stroke through various mechanisms; it is increasingly recognized as a cause of ischemic stroke with advanced imaging and is best managed with lifestyle modifications and medical therapy. The diagnostic search for the vulnerable culprit atherosclerotic plaque is an area of intense research, from the level of the intracranial arteries to that of the aortic arch. Ultrasonography and novel magnetic resonance imaging techniques (high-resolution vessel-wall imaging) may assist in the identification of vulnerable atherosclerotic plaques as the underlying cause in cryptogenic or misdiagnosed non-atherosclerotic ischemic stroke. Vertebrobasilar atherosclerotic disease is less common than carotid artery disease; thus, high-quality data on effective prevention strategies are scarcer. However, aggressive medical treatment is also the gold standard to reduce cerebrovascular disease located in posterior circulation.
Introduction
Atherosclerotic disease involving stenotic and non-stenotic atherosclerotic plaques of the extracranial and intracranial cerebral arteries as well as the aortic arch is a major cause of ischemic stroke. Regardless of the location of the culprit atherosclerotic plaque, atherosclerosis is a systemic disease affecting the brain, the heart, the aorta, the peripheral vessels, and the kidneys. As a result, atherosclerotic risk factors and interventions to reduce atherosclerotic vascular disease are common regardless of the end organ. Atherosclerosis starts at an early age with the deposition of foam cells in the intima, leading to fatty streaks that will eventually progress to atherosclerotic plaques through the deleterious effects of multiple vascular risk factors [1]. Physical inactivity, obesity, and unhealthy diet can lead to dyslipidemia, which is a major driving force of atherosclerosis [2]. Hypertension and diabetes damage the endothelium and accelerate atherosclerosis [3].
Major recent breakthroughs in atherosclerotic disease prevention and treatment in diverse fields of medical science are critical to stroke physicians for the optimal treatment of atherosclerotic stroke patients. Ten potentially modifiable risk factors (hypertension, regular physical activity, dyslipidemia, diet, obesity, psychosocial factors, smoking, cardiac causes, alcohol consumption, and diabetes mellitus) account for 90% of the population attributable risks of stroke [4]. Lifestyle modifications and intensive medical treatment have proven benefits and have evolved considerably in recent years, making the net clinical benefit of surgical or endovascular interventions for extracranial atherosclerotic disease less important. Endovascular interventions for intracranial atherosclerotic disease (ICAD) are currently reserved only for recurrent ischemic strokes despite best medical therapy (BMT).
Exciting results from aggressive cardiovascular risk factor (CVRF) modification studies through BMT promise marked reduction of the risk of atherosclerotic stroke—but at a price that may be prohibitive for individuals or societies of limited resources. On the other hand, measures by healthcare policymakers to implement widespread lifestyle modifications are lowcost interventions for the primary and secondary prevention of atherosclerotic stroke, which could obviate the need for polypharmacy.
The aim of this narrative review is to present recent medical breakthroughs in the primary and secondary prevention of atherosclerotic stroke through lifestyle modifications, novel treatments of modifiable risk factors for atherosclerosis, and optimized antithrombotic treatment, as well as to discuss the current role of interventions for symptomatic and asymptomatic extracranial or intracranial atherosclerotic plaques.
Lifestyle modifications
The promotion of a healthy lifestyle is the cornerstone for primary and secondary atherosclerotic prevention of stroke. Smoking is an independent risk factor for ischemic stroke, but the excess risk of stroke appears to be reduced 2 to 4 years after smoking cessation [5]. Physically active individuals have a 25% to 30% lower risk of stroke or mortality [6]. Healthy adults should perform moderate- to vigorous-intensity aerobic physical activity at least 40 min/day for 4 days/week. Obesity increases the risk of ischemic stroke by 22% for overweight individuals and 64% for obese individuals compared with normalweight individuals [7]. Above a body mass index of 25 kg/m2, there is a 40% increased stroke mortality for every 5 kg/m2 increase in body mass index [8].
The 2013 lifestyle management guidelines from the American Heart Association (AHA) advocate a dietary pattern that emphasizes the intake of vegetables, fruits, and whole grains and limits the intake of sweets, sugar-sweetened beverages, red meat, saturated fat, and trans-fat [9]. An energy-unrestricted Mediterranean diet supplemented by nuts (walnuts, hazelnuts, and almonds) or by extra-virgin olive oil reduces stroke risk [10]. In the recently published Dietary Patterns, Sodium Intake and Blood Pressure (DASH-Sodium) trial, 30 days of a combined DASH+low sodium (1,150 mg) diet, compared with 30 days of a typical “American” non-DASH+high sodium (3,450 mg) diet, dramatically lowered blood pressure (BP) for those with systolic blood pressure (SBP) ≥150 mm Hg by 21 mm Hg [2]; even normotensive patients witnessed a 5-mm Hg reduction. Data on community interventions of sodium substitution with potassium have indicated similarly impressive results [11]. Promoting public awareness and public health initiatives against sodium consumption could be a highly cost-effective strategy for reducing the burden of hypertension and atherosclerosis.
Socioeconomic factors are an important risk factor for cardiovascular disease (CVD) and a major obstacle to implementing lifestyle modifications for stroke prevention. Low-income areas and low individual household incomes were found to be independent predictors of a higher 10-year risk for CVD [12]. Even in high-income countries that have seen reductions in overall stroke risk in recent years, inequality gaps were not bridged. Lower educational level, low income, and living alone are all associated with an increased risk of stroke recurrence, despite adjustment for CVRFs [13].
Dyslipidemia
High levels of cholesterol increase ischemic stroke risk [14], whereas low levels may increase hemorrhagic stroke risk [15], which partly explains the paradox that there is no association between total cholesterol and stroke mortality [16]. Treatment with statins reduces the risk of stroke in patients with or at high risk for atherosclerosis [17] by 21%; this risk reduction has been associated with each 1 mmol/L (39 mg/dL) decrease of low-density lipoprotein (LDL) [18]. High-intensity statin treatment reduces non-fatal cardiovascular events [19] and leads to lower stroke incidence, even in healthy individuals with LDL <130 mg/dL and high-sensitivity C-reactive protein levels >2 mg/L [20]. Despite initial concerns emerging from the negative association of cholesterol levels and hemorrhagic stroke, statin treatment is no longer considered to increase the incidence of hemorrhagic cerebrovascular adverse events [21].
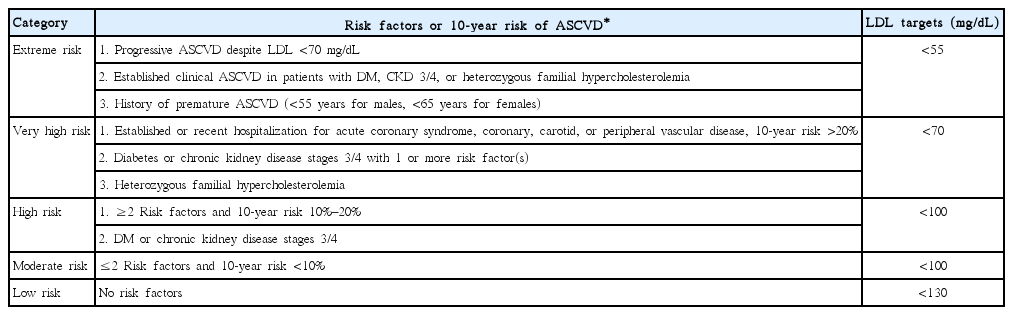
The first step in the medical management of dyslipidemia is to estimate total cardiovascular risk using a validated score; most risk estimation systems in use are based on either the Framingham [22] or the System for Cardiac Operative Risk Evaluation (SCORE) projects [23]. The 2013 American College of Cardiology (ACC)/AHA guideline on the Assessment of Cardiovascular Risk used the Pooled Cohort Equations derived from racially and geographically diverse cohort studies in order to estimate 10-year risk of atherosclerotic cardiovascular disease (ASCVD) [24]. The guidelines endorsed a previous statement from the AHA and American Stroke Association calling for the inclusion of ischemic stroke in the outcome of interest for CVD risk assessment [25]. A web-based application enabling estimation of 10-year and lifetime risk of ASCVD is available at http://my.americanheart.org/cvriskcalculator. The major determinant of an individual’s CVD risk is clinical ASCVD: acute coronary syndromes or a history of myocardial infarction (MI), stable or unstable angina, coronary or other arterial revascularization, stroke, transient ischemic attack (TIA), or peripheral arterial disease (PAD) presumed to be of atherosclerotic origin [26].
Since the announcement of the aforementioned guidelines, two randomized controlled trials (RCTs) have shown improved CVD outcomes with non-statin lipid-lowering medications: ezetimibe on top of simvastatin [27] and evocolumab on top of statin therapy [28]. Guidelines that incorporated these positive findings were published in 2017 and gave specific target LDL levels (Table 1) [29], following the approach described in an American expert consensus [30] and the European guidelines of 2016 [31]. These guidelines also recommend fibrates for patients with triglycerides >200 mg/dL and HDL <40 mg/dL. As we are awaiting the 2018 ACC/AHA guidelines, it seems that the updated LDL targets could be of benefit in atherosclerotic stroke patients [32]. An update on proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors has been recently published, which provides guidance for non-statin dyslipidemia treatment in order to attain very low LDL goals [33]. Add-on ezetimibe therapy may provide a 20% LDL level reduction, which may be insufficient for very high-risk patients [34]. PCSK9 inhibitors are recommended for very high-risk patients who—despite statins with or without ezetimibe therapy, or because of an inability to tolerate sufficient doses of at least three statins—do not attain the LDL goal of <140 or <100 mg/dL for those with rapidly progressive ASCVD or complex multivessel or polyvascular atherosclerotic disease. PCSK9 inhibitors are not indicated for asymptomatic ASCVD patients, except for patients with familial hypercholesterolemia who are at high or very high cardiovascular risk and have not attained LDL goals despite high-intensity statin plus ezetimibe therapy. It remains unclear how these guidelines will be implemented in clinical practice because the individuals eligible for treatment are substantially increased [35] and the cost-effectiveness of novel agents is hard to prove at the current price point [36]. When longer-term follow-up clinical data become available, treatment will likely have become cost-effective for very high-risk patients.
Unlike coronary heart disease and PAD, ischemic stroke has major non-atherosclerotic causes (cardioembolic and lacunar strokes). Therefore, it is sometimes challenging to prove etiology and leads to a high percentage of cryptogenic strokes despite extensive work-up, as will be discussed later in this article. In the seminal Stroke Prevention by Aggressive Reduction in Cholesterol Levels (SPARCL) study, the use of a high-intensity statin (80 mg of atorvastatin) for non-cardioembolic stroke/TIA patients without concomitant CVD led to reduced risk of fatal recurrent stroke by 43% and any cardiovascular event by 26% [37], proving that treatment of atherosclerosis in non-cardioembolic ischemic stroke has beneficial clinical implications. Stroke survivors had a 5-year vascular event risk of 17%, which is comparable to that of coronary risk equivalents. Even in the lowest risk group of 40- to 70-year-olds without coronary artery disease, the 5-year risk of MI or vascular death was 10% [38].
Atrial fibrillation (AF), the main cause of cardioembolic stroke, is also an independent predictor of MI or vascular event in stroke survivors. Recent research shows that AF patients with concomitant vascular disease are severely undertreated for vascular disease [39]. It should be kept in mind that the items included in the CHA2DS2-VASc score (used to calculate embolic risk in AF patients) are clinical expressions or risk factors of atherosclerotic vascular disease.
Consequently, comprehensive management of ischemic stroke patients should include implementation of the current guidelines for atherosclerotic vascular disease because they severely affect long-term outcomes. Using statin treatment guidelines for “presumably atherosclerotic” ischemic stroke patients means treating most stroke patients with high-dose statins (80 mg atorvastatin or 40 mg of rosuvastatin)—a practice supported by the highest quality evidence avaliable for statin treatment in secondary stroke prevention, the SPARCL trial. It should be noted that recurrent strokes during the SPARCL trial also tended to be less severe for patients on statins, a finding that has been since replicated [40]. Patients with atherosclerotic stroke pretreated with statins have greater neurological improvement 1 month later [41]. Patients who are pretreated with statins have lower prevalence of microembolic signals (MESs) on transcranial Doppler (TCD) monitoring, and the relationship between MES burden and statin pretreatment appears to be dose dependent [42].
Hypertension
Hypertension is a major risk factor for both ischemic and hemorrhagic stroke. Whereas hypertension is strongly linked to small-vessel cerebrovascular disease such as lipohyalinosis-related lacunar infarction, it is also a major cause of atherosclerotic stroke [43]. Throughout the range of BP, including normotensive values, a higher BP is associated with a greater risk of stroke [44]. Mean BP levels of 133/76 mm Hg, compared with 140/81 mm Hg, were associated with a reduction in risk of stroke by 22% in a meta-analysis of primary and secondary prevention studies [45]. The effect is even greater (at 27%) when only secondary prevention studies are considered: both SBP and diastolic BP reduction are linearly related to the lower risk of recurrent stroke, and aggressive BP control to a target of <130 mm Hg seems to be effective for secondary stroke prevention [46].
Recent guidelines have confirmed the importance of strict BP control and eliminated the term “pre-hypertension” by lowering normal BP levels to <120/80 mm Hg [47]. BP ranging from 130/80 to 140/90 mm Hg is now considered stage 1 hypertension and should be treated in all symptomatic ASCVD individuals and asymptomatic individuals with 10-year ASCVD risk over 10%; an exception is only made for secondary stroke prevention, for which the cutoff for antihypertensive treatment initiation of 140/90 mm Hg is maintained, but the BP goal for those treated remains at 130/80 mm Hg. The higher cutoff proposed for secondary stroke prevention is based on multiple observations that very low BP may harm stroke patients. A lower SBP target (<130 mm Hg) is recommended only after lacunar stroke; however, even in this context, the benefit seems to be derived from a reduced risk for hemorrhagic but not ischemic stroke [48]. SBP of >140 mm Hg in symptomatic patients with intracranial stenosis is associated with an increased risk of stroke [49]; however, low diastolic BP may be deleterious, especially when accompanied by increased pulse pressure of >60 mm Hg [50]. In a post hoc analysis of the Prevention Regimen for Effectively Avoiding Second Strokes (PROFESS) trial, low BP in patients after non-cardioembolic stroke was associated with worse outcomes; however, it could not be discerned whether low BP was causal or simply an indicator of poor health status [51].
Diabetes mellitus
Diabetic patients are at risk for both lacunar stroke [52] and large-artery stroke [53]. Diabetes may accelerate atheromatosis by inflammatory pathways through the production of free radicals and inhibition of nitric oxide synthase, exposing the endothelium to shear stress [54]. A 10-year duration of diabetes mellitus triples the risk for stroke, while one in five patients with diabetes mellitus will die of stroke [55]. Diabetic patients have both a high prevalence of CVRF and increased susceptibility to atherosclerosis. Lifestyle modifications and pharmacotherapy aim to accomplish glycemic control with a target glycated hemoglobin of <6.5% for most patients, while achieving lipid and BP targets is important for reducing ASCVD risk for patients with diabetes or prediabetes [56]. The Action to Control Cardiovascular Risk in Diabetes Blood Pressure (ACCORD BP) trial has demonstrated that intensive BP control (i.e., target SBP of <120 mm Hg as opposed to <140 mm Hg) reduces stroke risk by 41% in diabetic patients at increased ASCVD risk, at the expense of an increased risk of renal insufficiency (number needed to harm=50) [57]. Aspirin should be taken by diabetic patients at high risk of ASCVD for the primary prevention of CVD, although it is unclear whether it contributes to primary stroke prevention [58]; there is also an ongoing debate whether diabetic patients need a greater dose of aspirin because of an increased prevalence of resistance to aspirin [59].
Metformin remains the first-line treatment for type 2 diabetes mellitus patients according to guidelines, and metformin has a potential benefit for patients with clinical atherosclerotic disease [60]. When appropriate, diabetic patients with atherosclerotic stroke should also receive oral therapy with agents that have proven significant reduction in atherosclerotic vascular events in clinical trials. These agents are two sodium-glucose cotransporter 2 (SGLT2) inhibitors (empagliflozin [61] and canagliflozin [62]) and one glucagon-like peptide 1 (GLP-1) receptor agonist (liraglutide [63]), as well as one thiazolidinedione (pioglitazone [64]) that has also shown reduction in the progression of carotid atherosclerosis [65].
Antiplatelet therapy
Although anticoagulation remains the mainstay of thromboprophylaxis in cardioembolic stroke, antiplatelets are the preferred antithrombotic treatment for primary and secondary prevention of atherosclerotic stroke, even if the borders are becoming nuanced in most recent clinical research, as will be discussed later. Initiation of aspirin within 48 hours after ischemic stroke leads to a significant reduction in recurrence within 2 weeks [66]. The addition of dipyridamole appears to be slightly more effective [67] and clopidogrel appears to be equally effective on secondary stroke prevention when compared to aspirin plus dipyridamole [68]. Clopidogrel has been shown to be slightly more efficacious than aspirin for the composite outcome of ischemic stroke, MI, or vascular death [69]; this benefit is more pronounced for patients with polyvascular disease and those with symptomatic atherosclerotic disease [70]. It could also be an option for secondary stroke prevention after non-cardioembolic ischemic stroke while the patient was on aspirin [71].
The benefits of long-term treatment with aspirin plus clopidogrel (dual antiplatelet therapy [DAPT]) in patients with acute coronary syndrome were never replicated in stroke patients. Furthermore, DAPT has shown same efficacy but more bleeding complications than aspirin monotherapy [72,73]; more importantly, long-term DAPT after lacunar strokes is associated with increased mortality [74]. A Chinese study has shown that a 90-day regimen of DAPT after TIA or minor stroke reduces risk of recurrence [75]. A reduction of major ischemic events (with most of the prevented events being ischemic strokes) with DAPT has been replicated in Western populations. However, patients on DAPT also showed a higher risk of major hemorrhage (namely extracranial) at 90 days than those who received aspirin alone [76]. Nevertheless, DAPT was beneficial (compared to aspirin monotherapy) in reducing the composite endpoint of ischemic stroke, MI, or death from ischemic vascular causes, as well as the composite endpoint of ischemic and hemorrhagic stroke. Notably, secondary analysis of the results revealed that prevention of ischemic outcomes with DAPT was significant throughout the first 7 to 30 days of treatment, whereas the risk of major hemorrhage became greater only during the period from 8 to 90 days. For every 1,000 patients treated with DAPT, a total of 15 ischemic events would be prevented and five major hemorrhages would occur.
In patients with ischemic stroke or TIA with potentially symptomatic atherosclerotic stenosis of ipsilateral intracranial or extracranial arteries, ticagrelor plus aspirin reduced stroke recurrence compared to aspirin alone. A composite vascular endpoint (i.e., stroke, MI, or vascular death) occurred within 90 days for 6.7% of patients treated with ticagrelor, which was significantly less than the 9.6% in the aspirin arm of the trial [77]. It is worth mentioning that no difference of vascular endpoints was noted in patients without lacunar stroke, highlighting the fact that secondary thromboprophylaxis for atherosclerotic stroke should be studied in dedicated trials and that aggressive antiplatelet treatment seems to be a valid therapeutic strategy to reduce residual recurrence risk in atherosclerotic stroke patients. Another antiplatelet drug, cilostazol [78-80], was found to be non-inferior to aspirin in a recent clinical trial for the prevention of cardiovascular events (the composite of stroke, MI, or vascular death) in Asian patients who had ischemic stroke with a history of or imaging findings of intracerebral hemorrhage or two or more microbleeds; however, it did not reduce hemorrhagic events [81]. In an open-label part of the same trial, the addition of probucol to aspirin or cilostazol seemed to reduce the incidence of cardiovascular events.
Anticoagulation therapy
Apart from the positive results with aggressive antiplatelet therapy (either DAPT or ticagrelor), a breakthrough in 2017 was the publication of the Cardiovascular Outcomes for People Using Anticoagulation Strategies (COMPASS) trial. For the first time, a combination of an anticoagulant (2.5 mg of rivaroxaban twice a day) and aspirin was proven superior both to aspirin alone and to 5 mg of rivaroxaban twice a day in stable atherosclerotic disease [82]. This study included patients with different expressions of atherosclerosis: coronary, peripheral, and carotid artery disease that was either symptomatic (after intervention) or asymptomatic (>50% stenosis). A composite outcome of ischemic and hemorrhagic events was significantly in favor of patients in the combined treatment group. What is even more exciting is the fact that efficacy outcomes were mainly driven by a 50% relative risk reduction in ischemic stroke risk (P<0.001).
It should be stressed that a significant reduction in mortality was noted with a twice-daily 2.5-mg dose of rivaroxaban, as seen in a previous trial on acute coronary syndrome in which a twice-daily 2.5-mg dose of rivaroxaban on top of DAPT was superior to DAPT alone or DAPT plus a twice-daily 5-mg dose of rivaroxaban [83]. However, more bleeding was noted in the combined treatment group, even if fatal and intracranial bleeding were not significantly different. Therefore, rivaroxaban plus aspirin should be used with caution in patients who are at risk of bleeding, such as older patients and those with a history of gastrointestinal bleeding, taking into consideration the higher risk for gastrointestinal bleeding in AF-anticoagulation doses of rivaroxaban [84]. However, we could soon witness a very important change in the treatment guidelines on the secondary prevention of atherosclerotic stroke with the addition of twice-daily low-dose rivaroxaban to aspirin, changing the decadesold paradigm of antiplatelet therapy [85].
Anti-inflammatory therapy
Inflammation in atherosclerosis is an emerging therapeutic target. Data have shown that carotid plaque 18F-fluorodeoxyglucose (18F-FDG) uptake is an independent predictor of early stroke recurrence in patients with recently symptomatic carotid stenosis [86]. In the Canakinumab Antiinflammatory Thrombosis Outcome Study (CANTOS), canakinumab (a human monoclonal antibody against interleukin-1β) at a dose of 150 mg every 3 months was shown to reduce incidence of the composite outcome of non-fatal MI, non-fatal stroke, and cardiovascular death in patients after MI who had increased levels of highsensitivity C-reactive protein [87]. Hematologic side effects included neutropenia and thrombocytopenia; patients in the treatment arm also had increased risk for fatal infections. Mortality was equal in both arms because the patients on treatment had unexpectedly lower cancer-related death rates. The main limitation for clinical implementation is the vertiginous annual cost of treatment at 200,000 USD [88].
On the contrary, colchicine is a low-cost anti-inflammatory drug that does not carry a risk for fatal infections. Low-dose colchicine reduces the composite incidence of acute coronary syndrome, out-of-hospital cardiac arrest, or non-cardioembolic ischemic stroke in patients with stable coronary artery disease [89]. Colchicine could prove to have a significant role to play in plaque stabilization [90]; an RCT is ongoing [91].
Symptomatic extracranial carotid disease
There is robust evidence that carotid endarterectomy (CEA) or carotid artery stenting (CAS) of symptomatic internal carotid stenosis (ICA) of >70% on top of BMT is beneficial in the first 2 weeks after an ipsilateral non-disabling stroke or TIA with a number needed to treat (NNT) of 6 to prevent one major stroke at 2 years [92]. For patients older than 70 years, CEA seems to be preferable to stenting (Figure 1) [93]. A recently published pooled analysis of RCTs showed that CAS carries a significantly higher periprocedural risk compared to CEA, especially when the intervention is performed in the first 2 weeks following the index event [94]. This is exactly the time window of opportunity to maximize the benefit from intervention. However, postponing the intervention for 48 hours may be prudent, as there are concerns that ultra-early intervention during the first 2 days after the index event may carry increased risk [95]. There is a strong case for DAPT in the time interval between stroke and intervention, as clopidogrel load on top of aspirin therapy leads to emboli reduction [96] as measured by TCD and reduces recurrence by 40% with an excess of minor bleeding complications related to surgery [97].

Symptomatic extracranial internal carotid artery (ICA) atherosclerotic plaque: (A, B) ultrasound imaging, (C) computed tomography (CT) angiography, and (D) pathology specimen. A 52-year-old male smoker with history of hypertension presented with transient aphasia and right hemiparesis lasting for 24 minutes. He reported two similar transient episodes of neurological dysfunction during the last week lasting <10 minutes. Brain CT was normal. Carotid duplex showed a heterogeneous mostly hypoechogenic plaque causing a hemodynamically significant (>70%) stenosis of the left ICA (peak systolic velocity 229 cm/sec, end diastolic velocity 106 cm/sec, ICA/ common carotid artery [CCA] peak systolic velocity ratio 4.4). Ultrasound findings were confirmed on CT angiography. The patient received loading dose of clopidogrel (300 mg) and aspirin (100 mg) and underwent uneventfully carotid endarterectomy the next day of his hospital admission. He was discharged with no residual neurological symptoms on the fifth day of ictus.
The benefits from intervention diminish rapidly with time. Especially in women, no benefit was found when intervention was delayed more than 2 weeks after randomization [98]. CAS is a therapeutic alternative, especially in patients with severe medical comorbidities, challenging anatomy, prior neck operation or irradiation, post-CEA restenosis, or contralateral carotid occlusion. Periprocedural risk for CAS appears to be operator-dependent [99]. In any case and regardless of treatment modality, current guidelines insist that intervention is indicated only if the perioperative morbidity and mortality risk is estimated to be <6% [100]. The benefit-to-risk ratio is much less favorable for women [101]; sex-specific consensus states that perioperative risk should be <4% for women undergoing intervention in order to have robust clinical benefit [102]. It is obvious that this complication rate cutoff is low, but it is far from negligible. Given the fact that BMT has greatly evolved since the RCTs for CEA and CAS, conservative treatment has re-emerged as a valid option even in this high-risk patient group, especially for women, patients younger than 75 years, and cases in which intervention cannot be offered within 2 weeks of the index event [103].
Benefit is much less important after revascularization of a moderate (50% to 69%) symptomatic ICA stenosis. A total of 15 patients with moderate symptomatic ICA stenosis need to undergo CEA to prevent one ipsilateral stroke during a 5-year period [104]. It should be kept in mind that the aforementioned NNT is derived from a clinical trial that was conducted decades ago, when hypertension and dyslipidemia were less optimally treated compared to current clinical practice. On the other hand, technical advances in carotid interventions are numerous but have not shown a proven reduction in perioperative risk [105]. A reduction in perioperative risk in recent trials (going down to 1.4% in the Carotid Revascularization Endarterectomy Versus Stenting Trial [CREST]) [106] could be partially explained by the improvement of BMT that is always integrated in the intervention arm, especially statin treatment. Imaging findings that correlated with vulnerable plaques, which are found in patients who are presumably more likely to benefit from intervention, have shown that indices of plaque inflammation on 18F-FDG positron-emission tomography (PET)/computed tomography (CT) and high-resolution magnetic resonance imaging (HRMRI) were independent predictors of stroke recurrence [107]. Future studies including molecular imaging research [108] could identify high-risk groups who would be the most appropriate candidates for intervention. For the time being, intervention can be still justified in most patients with low predicted perioperative risk, especially males. Age could be used as a criterion for selecting the intervention type, because CEA seems to be more beneficial for patients older than 70 years [109].
Asymptomatic extracranial carotid disease
In the Framingham Heart Study population, the prevalence of >50% ICA stenosis was 7% in women and 9% in men older than 65 [110]. Current guidelines state that it is reasonable to perform CEA in asymptomatic patients who have >70% ICA stenosis if the risk of perioperative risk is lower than 3% (class IIa; evidence level A) and that CAS might be considered in “highly selected patients” (class IIb; evidence level B), but at the same time mention that the benefit over BMT is not well established [111]. It still remains elusive what criteria should be used to identify high-risk patients. There is good evidence that only a minority of patients on BMT for asymptomatic ICA stenosis will benefit from intervention in the current era of intense CVRF management [112]. Specifically, in 1995, the 5-year risk for ipsilateral stroke was 11% [113], reduced to 5.3% by 2004 [114] and just 3.6% by 2010 [115], which corresponds to an annual absolute risk of just 0.7% and to an overall relative risk reduction of 70% over these 15 years. In contrast to RCTs of symptomatic ICA stenosis, trials of asymptomatic stenosis revealed a limited number of patient characteristics that may predict stroke; for example, ICA occlusion carries a higher risk for stroke only when the contralateral ICA stenosis is symptomatic [116]. Male sex and age <75 years were associated with increased stroke risk [114]. A history of contralateral TIA or stroke appears also to be strongly associated with increased annual stroke risk [117]. Silent infarcts on brain imaging are also associated with subsequent stroke in asymptomatic ICA stenosis [117].
Data are still being accumulated to determine whether asymptomatic atherosclerotic plaques should be medically treated. The Objective Randomised Blinded Investigation with Optimal Medical Therapy of Angioplasty in Stable angina (ORBITA) study was published in 2018, which provided class I evidence that percutaneous coronary intervention (PCI) does not increase exercise time in patients with medically treated angina and severe coronary stenosis [118]. This negative result confirms the conclusion of a previous meta-analysis [119]. Despite the limitation of a small study group and a tendency for improved times in the PCI arm, it stands in stark contrast to current practice of performing a half-million PCI procedures for symptomatic relief of stable angina in the United States and Europe each year [120]. We are witnessing the paradox that good-quality data in favor of interventions comes from acute symptomatic atherosclerosis (carotid or coronary), but interventions in asymptomatic patients with stable atherosclerosis are far more numerous. The problem is much more important than some may think: 92% of all CEA and CAS in the United States were performed in asymptomatic patients in 2005 [121]! We are not far from having overwhelming evidence that BMT has outraced interventions for primary prevention of atherosclerotic disease [122]. Thus, there is a good chance we are exposing most asymptomatic atherosclerotic patients to unnecessary risk [123]. There is an urgent need to pinpoint the minority of asymptomatic patients with vulnerable atherosclerotic plaques that are at risk of provoking symptoms.
It is generally accepted that the main mechanism of stroke in the context of ICA stenosis is artery-to-artery embolism [124]. Vulnerable plaques that are prone to rupture and cause distal embolism can be detected with ultrasonography [125]. Plaque composition is assessed on B-mode with ultrasound and can be classified according to echogenicity as types I to V. Plaque surface can be smooth, irregular, or ulcerated [126]. A systematic review reported that plaque angiogenesis, complex plaque morphology, ulceration, echolucent plaques, and intraplaque motion were associated with symptomatic plaques [127]. There is evidence that heterogeneous plaques comprising of a mixture of hyperechoic, hypoechoic, and isoechoic lesions are significantly more prone to producing symptoms. Patients presenting with vulnerable plaques may be at higher risk for stroke and should be assessed for intervention. The Gray-Scale-Median (GSM) is a computerized measurement of the content of atheromatous plaques, in which low values correspond to hypoechoic plaques [128]. A GSM of <15 is associated with a higher stroke risk, but intervention for these plaques also carries a higher periprocedural risk [129].
Another method of ultrasonography with promising results for selecting high-risk patients is TCD detection of MES. Patients with MES on baseline TCD had an absolute annual risk of stroke ipsilateral to a >70% asymptomatic ICA stenosis of 3.7%, whereas patients without MES recording had only 0.7% annual stroke risk [130]. Approximately 10% of screened patients were MES positive; for the other 90% of patients, negative predictive value was high (94%), indicating that MES detection is a promising non-invasive technique to select patients at risk of stroke who would benefit from intervention. CT angiography has excellent accuracy for estimating ICA stenosis and is much more sensitive than ultrasonography for detecting plaque ulceration [131]. Fatty plaques appear hypoattenuated (<50 Hounsfield units [HU]), calcified plaques are hyperattenuated (>120 HU), and mixed plaques present with intermediate HU values [132]. Nevertheless, CT angiography for plaque characterization is limited by its low sensitivity to detect hemorrhage or a lipid core within atheromatous plaques. On the contrary, HRMRI can better depict plaque composition, especially intraplaque hemorrhage (IPH), which seems to be critical marker of plaque instability. IPH has been associated with a three-fold increase in the risk of stroke ipsilateral to ICA stenosis in asymptomatic individuals [133].
In conclusion, advances in medical treatment have raised questions regarding the benefit of CEA or CAS, especially for moderate symptomatic and asymptomatic ICA stenosis. Three RCTs are currently recruiting patients [134]. CREST-2 will separately test CEA and CAS for asymptomatic ICA stenosis versus BMT [135]. Asymptomatic Carotid Surgery Trial 2 (ACST-2) will directly compare CEA to CAS for asymptomatic ICA stenosis [136]. European Carotid Surgery Trial 2 (ECST-2) will also recruit patients with symptomatic stenosis [137]. The results of these trials will probably be available after 2020, at which time they will hopefully clarify the role of carotid interventions.
Non-stenotic symptomatic ICA plaques
In a pooled analysis of RCTs, it has been shown that intervention is of no benefit for symptomatic ICA stenosis of <50% [92]. Similarly, according to the Trial of Org 10172 in Acute Stroke Treatment (TOAST) classification, in order for an ischemic stroke to be considered due to large-artery atheromatosis (LAA), the culprit lesion has to cause a stenosis of >50% [138]. It is clear that there will be atheromatous plaques resulting in <50% stenosis that provoke distal embolism. These strokes/TIAs will be classified as cryptogenic if there is no concomitant stroke cause, but in fact they are missed LAA strokes. It has been suggested that luminal narrowing is not a prerequisite for plaque vulnerability [139] and it is known that most coronary artery plaques resulting in MI result in stenosis of <50% [140]. However, there has been limited availability of imaging techniques to visualize plaque content.
In a seminal study of black-blood MRI examination of the carotid arteries in patients with cryptogenic hemispheric stroke who presented with non-stenotic atherosclerotic plaques thicker than 2 mm in the carotid bifurcations, AHA type VI plaques (complicated plaques with IPH, thrombus, or rupture of the fibrous cap [141], known to be overrepresented in symptomatic stenotic ICA plaques [142]) were found exclusively on the ipsilateral side of stroke (37.5% vs. 0.0%) [143]. The results were replicated using a routine magnetic resonance (MR) angiography protocol performed on the day of stroke; intraplaque high-intensity signal, a marker of IPH, was once again exclusively found in non-stenotic plaques ipsilateral to the cerebral cryptogenic infarction (22% vs. 0%). Non-stenotic plaques of ≥5-mm thickness on CT angiography were present in 11% of ipsilateral and 1% of contralateral arteries in patients with embolic stroke of undetermined source (ESUS); plaques of ≥4-mm thickness were present in 19% and 5%, respectively, and plaques of ≥3-mm thickness were present in 35% and 15%, respectively [144]. This important difference of 20% could correspond to up to one in five ESUS patients having non-stenotic LAA stroke, which would likely not benefit from anticoagulation but would potentially benefit from strict CVRF modification, aggressive short-term antiplatelet therapy, and high-intensity statin treatment. A cross-sectional multicenter carotid MRI study of cryptogenic hemispheric stroke patients, besides revealing high-risk plaque characteristics more frequently on the symptomatic side, found maximal plaque thickness as a strong indicator of high-risk plaque imaging findings [145].
Carotid MRI in stroke-free participants in the Rotterdam study also reported increased IPH prevalence by quartile of plaque burden [146]. There have been many reports that challenged the strict TOAST criteria for LAA stroke; in the Causative Classification System of acute ischemic stroke, the term “probable atherosclerotic stroke” has been added [147]. In the Subtypes of Ischaemic Stroke Classification System, using a total plaque area (TPA) cutoff of ≥1.19 cm2 resulted in the detection of 33% of cases with LAA stroke versus 21% based on TOAST criteria [148]. This cutoff has been shown to be associated with a 5-year risk for stroke, death, or MI of 19.5% after adjustment for multiple risk factors [149]; interestingly enough, TPA was a stronger predictor of recurrent stroke risk than carotid stenosis [150]. Finally, a combined high-resolution carotid MRI and 18F-FDG PET—apart from confirming once again the presence of AHA type VI lesions ipsilateral to cryptogenic stroke (39% vs. 0 %, P=0.001)—revealed that patients harboring at least one type VI lesion showed significantly higher 18F-FDG uptake in both carotid arteries, which suggests that atherosclerosis is a diffuse inflammatory process [151].
Extracranial vertebral disease
There is evidence that LAA stroke is frequent in the vertebrobasilar circulation [152]. Ultrasonographic detection of >50% stenosis in the vertebral arteries is problematic because the origin of the vertebral artery is a common site of stenotic plaques in posterior circulation stroke [153]; however, in almost half of patients, it cannot be properly assessed with ultrasonography [154]. CT and contrast-enhanced MR angiography both have high sensitivity compared with digital subtraction angiography; CT angiography also presents high specificity [155]. The Carotid and Vertebral Artery Transluminal Angioplasty Study (CAVATAS) [156] was the first trial to examine the benefits from stenting stenotic atheromatous lesions; however, it only included 16 patients with posterior circulation. The phase 2 Vertebral Artery Stenting Trial (VAST) randomized 115 patients to either BMT or BMT with stenting [157]. The risk of stroke in the medically treated group was low, without a significant difference from patients after stenting; however, 5% of patients suffered from vascular death, MI, or any stroke within 30 days after intervention. In the Vertebral Artery Ischaemia Stenting Trial (VIST), 182 posterior circulation stroke patients were randomized to either BMT plus stenting or BMT; despite the fact that there were no periprocedural complications with extracranial vertebral artery stenting, very low recurrence rates of posterior circulation stroke under BMT precluded any significant benefit from intervention [158]. Vertebral intervention should thus be reserved only for those rare cases of recurrence despite BMT [100].
As opposed to LAA strokes in the anterior circulation, little is known about the risk stratification for recurrence in patients after vertebrobasilar stroke/TIA. A prospective observational trial evaluated distal blood flow compromise as measured by large-vessel quantitative MR angiography in patients with LAA stroke/TIA of the posterior circulation [159]. Low distal flow status was found in 25% of patients and was associated with an increased risk for vertebrobasilar stroke recurrence compared to patients with normal distal flow [160]. This low distal flow group appears to be at particularly high risk for recurrence when BP is <140/90 mm Hg; this is an important finding that needs further research because one of the major components of BMT for LAA stroke patients is strict hypertension control [161]. Nevertheless, it would be premature to discuss permissive hypertension for these patients because this approach has already been tried in stroke patients with intracranial stenosis, for whom mean SBP of ≥140 mm Hg led to higher rates of stroke, MI, and vascular death [162].
Aortic atherosclerosis
The association between complex (thickness >4 mm, presence of ulcers or mobile components, lack of calcification) [163] atheromatous plaques of the aortic arch and stroke has been described in autopsy [164], case-control [165], and prospective studies [166]. MES on TCD are frequent in complex aortic atheromatous plaques [167]. Some studies have challenged the causal link between aortic atheroma and stroke, asserting that aortic plaques are no more than a marker of generalized atherosclerosis [168] or fortuitous findings [169]. On the contrary, studies have also incriminated the descending aorta for harboring atheromatous plaques that may provoke brain embolisms via retrograde diastolic flow as seen on multidirectional three-dimensional (3D) MRI velocity mapping [170]. In addition, a higher prevalence of descending aorta complex plaques has been reported with cryptogenic stroke, suggesting an etiopathogenic link [171].
A systematic review of transesophageal echocardiography (TEE) studies found no association between aortic arch atherosclerosis and cryptogenic stroke [172]. However, TEE may yield a lower number of plaques in the aortic arch compared both to CT angiography [173] and 3D MRI [174]. Additional studies are necessary to prove that descending aorta atherosclerotic plaques can actually provoke brain embolism, especially when they are complex in the distal aortic arch and the proximal segment of descending aorta, where retrograde diastolic flow to brain arteries is maximal [175]. As for every site of atherosclerosis, statin therapy is crucial for aortic plaque regression [176] and inflammation reduction [177].
Intracranial atherosclerotic disease
ICAD is another expression of systemic atherosclerosis, although, until recently, its importance has been underscored compared with extracranial atherosclerotic disease. However, ICAD is a frequent cause of stroke and merits dedicated study because it differs from extracranial atherosclerosis. First of all, ICAD is more prevalent in Asian populations, but it remains unclear whether the cause is genetic or environmental [178]. The role of the environment seems to gain in importance because recent studies have reported that the westernization of lifestyles in Asia increased extracranial atherosclerotic disease prevalence [179]. Secondly, ICAD is unique in the multiplicity of mechanisms that provoke stroke: artery-to-artery embolism (Figure 2), branch disease (Figure 3), thrombosis, and hemodynamic impairment have been implicated in brain infarction from intracranial atherosclerotic stenosis in imaging studies [180]. MES detection with TCD, suggesting artery-to-artery embolism, has been in reported in 22% of symptomatic ICAD stroke patients [181]; this percentage climbs to 50% for severe stenosis [182], suggesting that arterial embolism could be the major cause of stroke in ICAD. MRI studies in Asian populations reported that 60% of strokes in ICAD arise from embolism, 15% from branch infarction, 14% from in situ occlusion, and less than 1% from hemodynamic impairment [3].

Symptomatic intracranial non-stenotic atherosclerotic plaque. A 67-year-old male with hypertension and diabetes mellitus, presented with acute-onset diplopia. On neurological examination Parinaud syndrome and incomplete left internuclear opthalmoplegia was identified. (A) Diffusion-weighted imaging showed two acute left mesencephalic ischemic lesions (oval). (B) Time-of-flight showed a focal stenosis of the distal basilar artery (asterisk). (C) 3-Tesla high-resolution vessel wall-imaging disclosed a diffuse uptake of gadolinium by the atherosclerotic plaque in the wall of the basilar artery indicative of active inflammation (arrows). Artery-to-artery embolism to distal paramedian midbrain branches of an unstable non-atherosclerotic plaque in distal basilar artery was considered the underlying mechanism of acute cerebral ischemia. The patient was treated with dual antiplatelet therapy (clopidogrel load of 300 mg followed by clopidogrel 75 mg once a day and aspirin) for 3 months and colchicine (0.5 mg once a day; off-label use for inflammatory atherosclerosis). Dual antiplatetet therapy was later substituted with single antiplatelet therapy (clopidogrel 75 mg). The patient experienced no recurrence during the 6-month follow-up period.
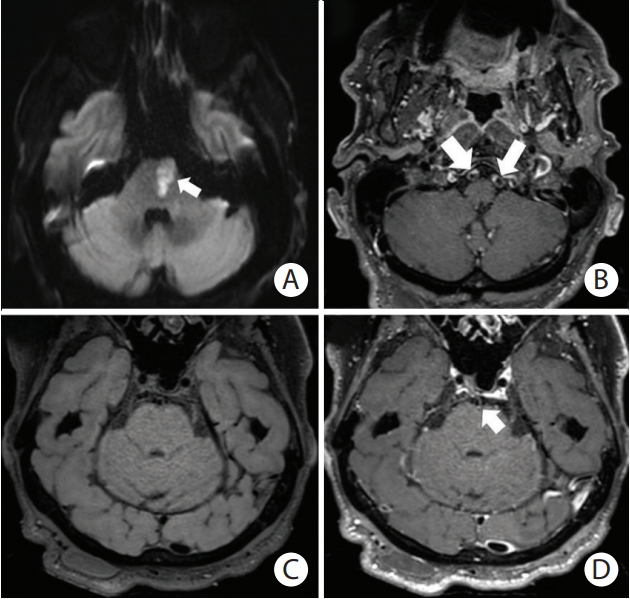
Branch-occlusive disease of the pons. (A) Diffusion-weighted image showing a left medial pontine infarction (arrow) along the left paramedian pontine artery territory (penetrating branch of basilar artery). (B) Post-contrast T1-weighted SPACE (Sampling Perfection with Application optimized Contrast using different angle Evolutions) image showing mural enhancement of both vertebral arteries (arrows). Patchy, eccentric pattern of contrast enhancement is consistent with atheromatous lesions. (C) Preand (D) post-contrast T1-weighted SPACE images showing uneven mural enhancement of the basilar artery (D, arrow) indicating atheromatosis at the level of the pons.
In the Warfarin versus Aspirin for Symptomatic Intracranial Disease (WASID) trial, stenosis of >70% doubled the risk of stroke recurrence compared to symptomatic stenosis of <70%; in fact, the risk increased linearly with stenosis severity, peaking at a 30% annual risk of recurrence for symptomatic stenosis of 80% to 89% and then declining for >90% stenosis [183]. The Chinese Intracranial Atherosclerosis (CICAS) study confirmed that the degree of stenosis correlates with recurrence risk; however, the correlation was moderate and other factors, such as a complete circle of Willis, seemed to play a more important role [184].
Atherosclerotic plaques resulting in stenosis of <50% can also lead to stroke. Branch atheromatous disease results in occlusion of the orifices of penetrating branches of the middle cerebral or basilar artery; it is commonly misdiagnosed as small vessel disease (lacunar stroke) [185]. MRI with HR vessel wall imaging has been used to reveal this elusive cause of stroke, which is usually related to non-stenotic atherosclerotic plaques that cannot be detected with conventional lumenography-based imaging modalities (Figure 3) [186]. Up to 36% of deep infarcts of the lenticulostriate arteries and 60% of infarcts involving the anterior pontine arteries group in Asians have been attributed to ICAD [187]. Advanced imaging with stronger magnetic fields may identify even more intracranial atheromatosis [188].
HR MRI is an increasingly valuable tool because it can provide data on intracranial plaque vulnerability [189]. IPH has been described in symptomatic plaques in postmortem studies [190] and has been frequently found as high signal in T1 MRI in symptomatic plaques [191]. Lipid component, plaque morphology, and neovasculature are plaque characteristics that seem to indicate increased stroke risk; further development of imaging methods may help to identify high-risk individuals who may benefit from aggressive treatment [192]. As with any site of atherosclerotic disease, stroke risk is much lower for asymptomatic ICAD in stroke patients whose workup revealed intracranial stenosis irrelevant to the index event [193]. There are limited data on the risk of ICAD in healthy individuals; however, it seems that asymptomatic ICAD is an independent risk factor for stroke but carries a low risk of becoming symptomatic, suggesting that aggressive global reduction of CV risk may be more appropriate than local interventions targeted to the asymptomatic plaque [194]. In the WASID trial, there was no difference in stroke recurrence between the anterior and the posterior circulation; however, in the CICAS study, posterior circulation ICAD stroke carried an increased risk of recurrence. A recent autopsy study showed that atherosclerosis is less frequent in the posterior circulation arteries and presents preferentially outward remodeling (resulting less frequently in focal stenosis) compared to anterior circulation arteries [195]. An overview of risk factors for stroke associated with ICAD is shown in Table 2 [49,162,181-184,190-194,196-208].
Regardless of stroke mechanisms, plaque location, or characteristics, ICAD is the expression of a single disease: atherosclerosis. There is clear evidence that the best secondary prevention strategy for ICAD stroke is aggressive BMT. An LDL of <70 mg/dL, SBP of <140 mm Hg (or <130 mm Hg in diabetic patients), and a comprehensive lifestyle modification program resulted in 12% yearly risk of stroke or death for symptomatic ICAD of >70% in the Stenting and Aggressive Medical Management for the Prevention of Recurrent Stroke in Intracranial Stenosis (SAMMPRIS) trial [192]. Medical treatment consisting of DAPT, one drug from each major class of antihypertensive agents, and high-intensity statin therapy led to much lower recurrence rates compared with the preceding WASID [183], in which patients in the single antiplatelet (aspirin) arm received standard risk factor management and the 1-year rate of stroke or death from vascular causes was 25%. Short-term (<90-day) DAPT for acute ICAD stroke is considered reasonable in current guidelines (class IIb; evidence level B). DAPT in ICAD stroke patients resulted in significant MES reduction as recorded on TCD within days of treatment [209]. However, the low recurrence rates for patients on DAPT have not been replicated in a prospective registry [210] or in a post hoc analysis of a RCT with ICAD patients [211].
Percutaneous transluminal angioplasty and stenting (PTAS) for symptomatic ICAD is associated with adverse periprocedural and long-term cerebrovascular events, both ischemic and hemorrhagic. PTAS was associated with a higher risk of recurrent ischemic stroke in the territory of qualifying vessel both within 30 days and 1 year, a higher risk of any ischemic stroke within 30 days from stroke, and a higher risk for intracranial hemorrhage both within 30 days and 1 year; furthermore, the composite outcome of any stroke or death within 1 year and 2 years was higher in PTAS than in medical therapy [212]. Perforator occlusion is likely causal for three out of four periprocedural ischemic strokes, presumably due to plaque debris being displaced or “snowplowed” over the perforator vessels during angioplasty or stent deployment [213]. Apart from the procedure-related ischemic stroke, the risk of cerebral ischemia in the territory of the stenotic artery remained increased 30 days post-intervention. Finally, an unexpectedly high risk of intracranial hemorrhage after stenting was consistently documented in RCTs, especially in severe stenosis with small vessel diameter; this was related to vessel perforation (resulting in subarachnoid hemorrhage) and reperfusion injury, periprocedural anticoagulation, and clopidogrel load in patients with severe hemodynamic impairment. An ongoing multicenter RCT in China will include ICAD TIA and stroke patients not earlier than 3 weeks and no later than 12 months after the index event caused by >70% stenosis of a major intracranial artery with a 2- to 4-mm diameter confirmed by digital subtraction angiography [214]. Patients will be randomized to either BMT or BMT plus stenting with a self-expanding Wingspan device. Until the publication of positive outcomes in RCTs, PTAS is not indicated for symptomatic ICAD stenosis unless there is recurrence despite BMT.
Conclusions
The main current updates in the prevention of atherosclerotic stroke include the potential of anti-inflammatory agents to offer additional risk reduction on top of BMT consisting of BP, blood glucose, and blood cholesterol control as well as optimal antiplatelet therapies and lifestyle modifications. Aggressive LDL reduction with high-dose statins coupled with ezetimibe and/or PSCK9 inhibitors further reduce the risk of cardiovascular events in patients with stroke or MI. The combination of low-dose anticoagulation (2.5 mg of rivaroxaban twice daily) and antiplatelet (100 mg of aspirin) therapies appears to be superior to single antiplatelet or anticoagulation therapy for the secondary prevention of patients with stable carotid artery disease. Carotid and coronary revascularization procedures appear to offer little or no supplementary benefit for patients with stable (or asymptomatic) coronary or carotid artery disease compared to BMT. High-resolution vessel-wall imaging MRI may provide a reliable, non-invasive neuroimaging marker of unstable atherosclerotic plaque in extracranial and intracranial vessels.
The main current updates in the prevention of atherosclerotic stroke include the potential of anti-inflammatory agents to offer additional risk reduction on top of BMT consisting of BP, blood glucose, and blood cholesterol control as well as optimal antiplatelet therapies and lifestyle modifications. Aggressive LDL reduction with high-dose statins coupled with ezetimibe and/or PSCK9 inhibitors further reduce the risk of cardiovascular events in patients with stroke or MI. The combination of low-dose anticoagulation (2.5 mg of rivaroxaban twice daily) and antiplatelet (100 mg of aspirin) therapies appears to be superior to single antiplatelet or anticoagulation therapy for the secondary prevention of patients with stable carotid artery disease. Carotid and coronary revascularization procedures appear to offer little or no supplementary benefit for patients with stable (or asymptomatic) coronary or carotid artery disease compared to BMT. High-resolution vessel-wall imaging MRI may provide a reliable, non-invasive neuroimaging marker of unstable atherosclerotic plaque in extracranial and intracranial vessels.
Notes
Disclosure
The authors have no financial conflicts of interest.
Acknowledgements
Dong-Eog Kim was supported by Global Research Lab (GRL) program (NRF-2015K1A1A2028228) of the National Research Foundation, funded by the Korean government, Republic of Korea.