Recanalization before Thrombectomy in Tenecteplase vs. Alteplase-Treated Drip-and-Ship Patients
Article information
Dear Sir:
The Extending the time for Thrombolysis in Emergency Neurological Deficits–Intra-Arterial using Tenecteplase (EXTEND-IA TNK) trial recently showed 2-fold higher early recanalization (ER) rate before mechanical thrombectomy (MT) following intravenous thrombolysis (IVT) with tenecteplase 0.25 mg/kg, as compared to alteplase 0.9 mg/kg [1]. However, most included patients were directly admitted to MT-capable centres (‘mothership’ paradigm), implying short IVT-to-MT delays. Tenecteplase may therefore be preferred in the mothership setting. Here, we assessed ER rate before MT following tenecteplase or alteplase in patients transferred for MT from a non-MT-capable centre (‘drip-and-ship’ paradigm), i.e., implying longer IVT-to-MT delays, currently the most frequent situation [2].
Inclusion criteria for the present retrospective study were (1) acute stroke with large vessel occlusion treated with IVT with tenecteplase 0.25 mg/kg or alteplase 0.9 mg/kg; and (2) ER evaluation ≤3 hours from IVT start on pre-MT first angiographic run or non-invasive vascular imaging. Tenecteplase patients were all from one large French non-MT-capable centre, which based on previous trials [3,4] and for practical convenience opted to use tenecteplase off-label before transfer for MT. Alteplase patients were from 23 other French non-MT-capable centres. ER was defined as modified thrombolysis-in-cerebral-infarction scale ≥2b score. In accordance with French legislation, patients were informed of their participation in this study, and offered the possibility to withdraw. As per current French law, approval by an Ethics Committee was not required as this study implied retrospective analysis of anonymized data. To reduce the effects of potential confounders, a 1:1 propensity-score matching of patients from the tenecteplase group to patients from the alteplase group was performed, using confounders based on available literature [5].
From May 2015 to October 2017, 816 patients were identified (n=160 and n=656 tenecteplase- and alteplase-treated, respectively). In the propensity-score matched cohorts (n=131 per group), the main confounders for ER were well balanced (Table 1). ER occurred in 21.4% (95% confidence interval [CI], 14.4 to 28.4) versus 18.3% (95% CI, 11.7 to 24.9) patients from the tenecteplase- and alteplase-treated cohorts, respectively (odds ratio, 1.25; 95% CI, 0.65 to 2.41; P=0.51). There was no significant association between thrombolytic agent used and 3-month functional independence (modified Rankin score [mRS] 0 to 2: 56% vs. 56% in the tenecteplase- and alteplase-treated cohorts, P=0.75).
Comparing our study to EXTEND-IA TNK, ER rates following tenecteplase were similar (21% vs. 22%, respectively), but were markedly higher following alteplase (18% vs. 10%, respectively) [1]. The radically different care paradigm between the two studies, namely 100% drip-and-ship in our study versus 75% mothership in EXTEND-IA TNK [1], which translates into longer IVT-to-angiography delays (median: 93 minutes vs. 55 minutes, respectively), may account for the higher ER rate with alteplase in our study. Indeed, short IVT-to-angiography time implies that some patients, particularly with the mothership paradigm, do not receive the full alteplase dose before MT. Taken together with EXTEND-IA TNK, therefore, our data suggest that although in dripand-ship patients the recanalization rate before thrombectomy may be similar with both thrombolytics, recanalization may occur earlier with tenecteplase (Figure 1). In support, one study reported earlier recanalization with tenecteplase than with alteplase in a rabbit carotid thrombosis model [6]. If this hypothesis is confirmed, this may have clinical relevance given the strong relationship between timing of reperfusion and functional outcome. The lack of difference in 3-month mRS between the two thrombolytic agents in our study may be because any difference in recanalization timing would only concern approximately one in five patients, which may not translate into better functional outcomes across the whole sample.
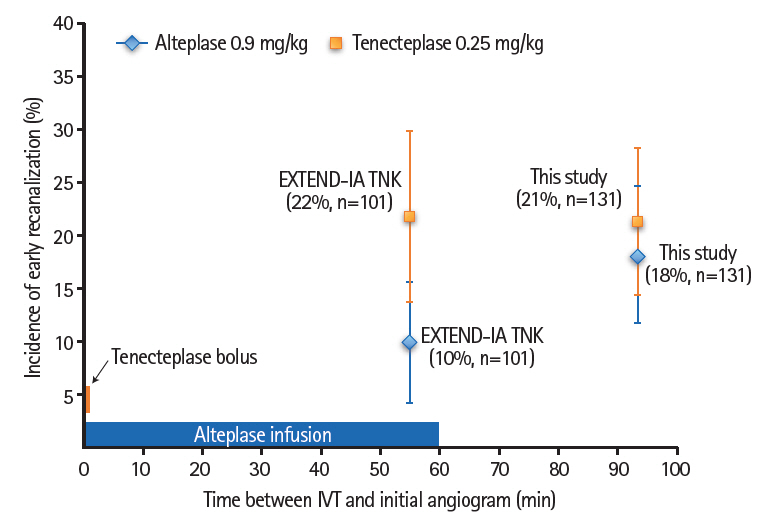
Association between early recanalization (ER) rate and time elapsed between intravenous thrombolysis (IVT) start and ER assessment in the present study and the Extending the time for Thrombolysis in Emergency Neurological Deficits–Intra-Arterial using Tenecteplase (EXTEND-IA TNK) trial. The red and blue bars in the lower left corner represent the duration of tenecteplase or alteplase intravenous administration. Red and blue squares represent ER incidence following tenecteplase and alteplase, espectively. Bars represent the 95% confidence intervals.
Our study has limitations. First, uncovered confounding factors cannot be ruled out, especially since the tenecteplase and alteplase groups were treated in different centers. Second, as the participating centers mostly used magnetic resonance imaging for patient workup, the population studied might differ from primarily computed tomography-assessed populations.
Notes
Pierre Seners is funded by la Fondation pour la Recherche Médicale (FDM 41382) and les Journées de neurologie de langue française (JNLF).
List of collaborators: Marie Abrivard, Sonia Alamowitch, Wagih BenHassen, Lise-Prune Berner, Yves Berthezene, Karine Blanc-Lasserre, Anne Boulin, Grégoire Boulouis, Stéphane Bouly, Frédéric Bourdain, David Calvet, Vladimir Charron, Mohamed Chbicheb, Stéphanie Condette-Auliac, Ovide Corabianu, Charlotte Cordonnier, Oguzhan Coskun, Jean-Philippe Cottier, Cyril Dargazanli, Thomas De Broucker, Jean-Pierre Decroix, Federico Di Maria, Sylvie Dorocant, Xavier Ducrocq, Serge Evrard, Hilde Henon, Isabelle Girard, Thibault Lalu, Laurence Legrand, Patrick Le Coz, Morgan Le Guen, Olivier Ille, Xavier Leclerc, Didier Leys, Christophe Magni, Eric Manchon, Mikaël Mazighi, François Mounier-Vehier, Marinette Moynier, Ioan-Paul Muresan, Norbert Nighoghossian, Michael Obadia, Elodie Ong, Canan Ozsancak, Frédéric Philippeau, Fernando Pico, Georges Rodesch, Thierry Rosolacci, Denis Sablot, Philippe Tassan, Maya Tchikviladze, Marie Tisserand, Francis Turjman, Anne-Evelyne Vallet, Adrien Wang, Marc Zins, and Mathieu Zuber.
The authors have no financial conflicts of interest.
