Optimizing Stem Cell Therapy after Ischemic Brain Injury
Article information
Abstract
Stem cells have been used for regenerative and therapeutic purposes in a variety of diseases. In ischemic brain injury, preclinical studies have been promising, but have failed to translate results to clinical trials. We aimed to explore the application of stem cells after ischemic brain injury by focusing on topics such as delivery routes, regeneration efficacy, adverse effects, and in vivo potential optimization. PUBMED and Web of Science were searched for the latest studies examining stem cell therapy applications in ischemic brain injury, particularly after stroke or cardiac arrest, with a focus on studies addressing delivery optimization, stem cell type comparison, or translational aspects. Other studies providing further understanding or potential contributions to ischemic brain injury treatment were also included. Multiple stem cell types have been investigated in ischemic brain injury treatment, with a strong literature base in the treatment of stroke. Studies have suggested that stem cell administration after ischemic brain injury exerts paracrine effects via growth factor release, blood-brain barrier integrity protection, and allows for exosome release for ischemic injury mitigation. To date, limited studies have investigated these therapeutic mechanisms in the setting of cardiac arrest or therapeutic hypothermia. Several delivery modalities are available, each with limitations regarding invasiveness and safety outcomes. Intranasal delivery presents a potentially improved mechanism, and hypoxic conditioning offers a potential stem cell therapy optimization strategy for ischemic brain injury. The use of stem cells to treat ischemic brain injury in clinical trials is in its early phase; however, increasing preclinical evidence suggests that stem cells can contribute to the down-regulation of inflammatory phenotypes and regeneration following injury. The safety and the tolerability profile of stem cells have been confirmed, and their potent therapeutic effects make them powerful therapeutic agents for ischemic brain injury patients.
Introduction
Decades of experimental evidence have shown the safety and efficacy of stem cell therapies in stroke animal models [1]. Despite promising outcome data from small, prospective, and phase I clinical studies [2,3], functional neurological outcomes from large randomized stem cell clinical trials have remained insufficient for supporting stem cell therapy in stroke patients [1], thus leaving a frustrating disconnection between the efficacy outcomes in preclinical versus clinical trials [4,5]. This inconsistency suggests that many factors that remain unknown in ischemic brain injury may impact the stem cell effects [1]. Translation of stem cell therapies from lab to patients requires a critical understanding of the different stem cell types and their mechanisms of action (i.e., cell replacement, growth factor secretion, exosome production, or inflammation regulation) [6-8], as well as an in-depth exploration of the ideal route, timing of administration, and cellular modification. These factors need to be optimized for acute ischemic stroke and ischemic brain injury treatment.
Exogenous stem cell transplantation can improve the outcomes of patients with stroke [8-15] and hypoxic-ischemic brain injury induced by cardiac arrest [16-18]. Stem cell therapeutic benefits are not limited to their ability to replace diseased cells and tissues, but also extend to the provision of a supportive neurogenesis microenvironment over an extended period of time [19] through bioactive neurotrophic factor secretion [20], mitochondrial transfer [8,21], and exosome release [7,22-25]. Transplanted stem cells can also be used as delivery vectors for agents such as chemotherapeutics, pro-drugs, and cytokines that can mediate inflammatory responses [26-28].
Despite the disappointing randomized controlled trial results, stem cell therapy remains a highly promising method to treat ischemic brain injury. Ischemic brain injury is defined as any injury to the brain caused by oxygen deprivation, such as ischemic stroke, perinatal hypoxic-ischemic encephalopathy (HIE), and cerebral ischemia secondary to cardiac arrest. We aim to summarize the work completed thus far, investigating stem cell sources, delivery routes, and methods for optimizing stem cell therapy, including survival and adhesion optimization. We will analyze this disconnection between preclinical and clinical outcomes, focusing on future directions for the clinical translation of this therapeutic intervention to promote its clinical application in ischemic brain diseases.
Stem cell sources
Stem cells belong to four major categories based on their differentiation potential (Figure 1) [29]. Totipotent cells, such as zygotes and early blastomeres, can generate the whole organism and represent a subset of stem cells with a huge differentiation potential; however, to date, these have not been successfully isolated and cultured in vitro [30]. Pluripotent cells, such as embryonic stem cells (ESCs) and induced pluripotent stem cells (iPSCs), can differentiate into any tissue-specific adult body cell [30,31]. Adult stem cells are multipotent and can differentiate into cell types within one particular lineage [31,32]. Recently, unipotent stem cells, such as spermatogonial stem cells, have been found to be an important tissue maintenance and cellular regeneration source [33].
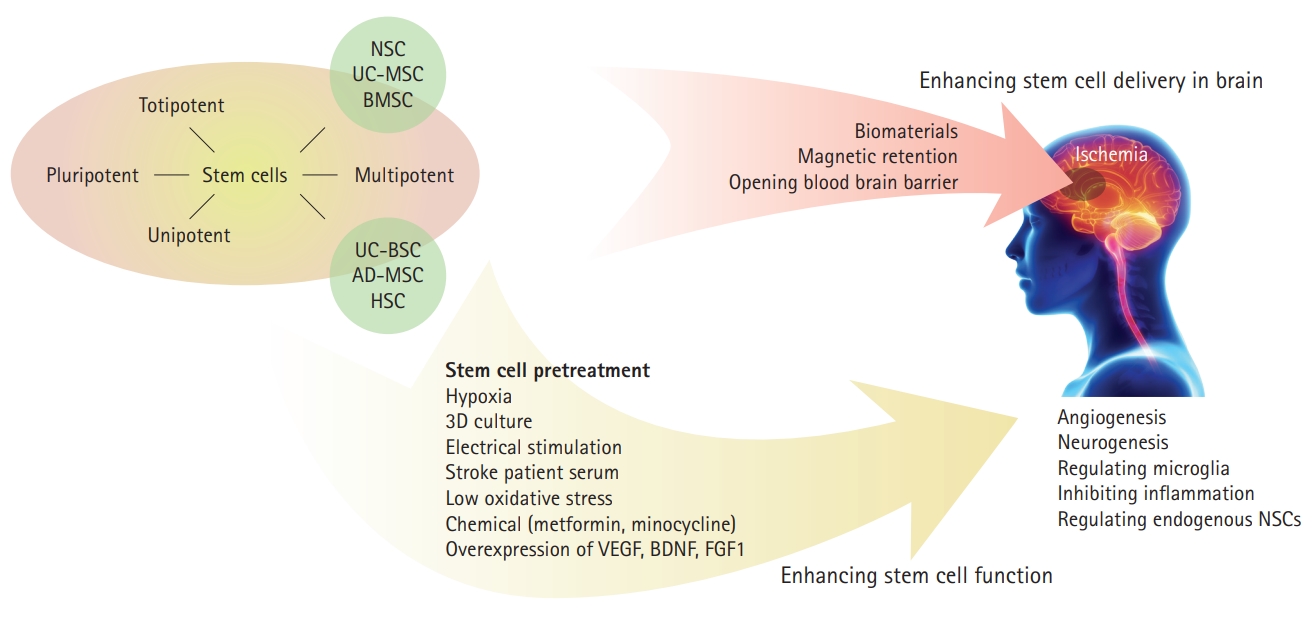
Strategy for improving stem cell application in ischemic brain injury. NSC, neural stem cell; UC-MSC, umbilical cord-derived mesenchymal stem cell; BMSC, bone marrow stem cell; UC-BSC, umbilical cord blood stem cell; AD-MSC, adipose-derived mesenchymal stem cell; HSC, hematopoietic stem cell; 3D, three-dimensional; VEGF, vascular endothelial growth factor; BDNF, brain derived neurotrophic factor; FGF1, fibroblast growth factor type 1.
Pluripotent stem cells
ESCs and iPSCs have a powerful renewal potential and can be manipulated to differentiate into phenotypes from all three embryonic layers [34]. Human iPSCs (hiPSCs) have been used for therapeutic solutions regarding central nervous system (CNS) disorders [35], although their use for ischemic brain injury has remained limited. This cell type has been limited in translational use due to a potential risk for insertional mutagenesis or oncogenesis, production of immune-tolerable cells, and a poor integration into host circuits [34,36].
Neural stem cells
Endogenous neural stem cells (NSCs) are primarily located in the sub-ventricular zone and dentate gyrus. Exogenous NSCs are extracted from three main sources for therapeutic purposes: direct extraction from brain tissue (particularly paraventricular or olfactory bulb regions), differentiation from pluripotent stem cells, and trans-differentiation from somatic cells, such as skin fibroblasts, renal tubular cells in urine, and blood cells [37]. Initially, human NSCs held constraints: limited donor availability, low proliferation rate, and poor long-term in vitro expansion. It has recently been possible to culture them over several passages and successfully differentiate them into neuronal and glial cell types [38].
Exogenous NSCs can migrate into ischemic brain areas, where they can differentiate into neurons or astrocytes and promote endogenous NSC proliferation and differentiation [39-41]. Administration of human NSCs in a stroke rat model revealed neuroprotective effects by increasing dendritic branching, enhancing corticospinal tract projections, and inhibiting inflammation [42,43]. Additionally, an in vitro and in vivo study has suggested that improved functional outcomes in stroke rats treated with transplanted NSCs may be associated with angiogenesis stimulation and brain microvasculature formation, due to the increased expression of proangiogenic factors in NSCs [44].
A small phase I translational approach demonstrated that ipsilateral implantation of CTX0E3 human NSCs into the putamen of chronic stroke patients was safe, with no adverse events related to stem cell administration after 2 years of follow-up, and was associated with a slight improvement in the National Institutes of Health Stroke Scale (NIHSS) [45]. Notably, adverse events related to the invasive surgery required for therapy were reported.
Despite the promising utility of NSCs, some limitations exist. A very high cell dose is required for transplantation. Tissue-derived NSCs can clot in vivo, if transplanted at a high density, and may be prone to a poor survival rate [37]. Further, our understanding remains limited about the molecular mechanisms regulating endogenous NSCs for neurogenesis and methods for enhancing endogenous neurogenesis in ischemic brain injury patients [46]. More work is needed to optimize NSC application conditions prior to clinical practice.
Mesenchymal stem cells
Mesenchymal stem cells (MSCs) can be derived from a variety of sources, but are primarily harvested from the adipose tissue (AD-MSCs) or bone marrow (BM-MSCs) [47]. BM-MSC harvesting requires an invasive procedure and results in relatively low stem cell yields, whereas large quantities of AD-MSCs, which are easily accessible, abundant, and have little ethical concerns regarding their use, can be harvested using less-invasive liposuction techniques [20,48]. MSCs are the most studied stem cell in stroke treatment, providing robust information for guiding stem cell application.
Preclinical studies demonstrate that transplantation of MSCs in an early stage of stroke improves neurological functional outcomes by regulating microglia phenotypes and stimulating regenerative processes [49,50]. In cardiac arrest-induced brain injury, MSC administration, together with therapeutic hypothermia, can markedly enhance neuroprotection and reduce hippocampal neuronal death [51]. Unfortunately, MSCs are prone to accumulation and aggregation in other organs, particularly the lungs (up to 80% of cells), after intravenous (IV) administration, which may result in embolism or hemorrhage [52,53].
Adipose-derived MSCs
AD-MSCs display a homing tendency toward sites of brain injury, with neuroprotective effects in ischemic brain injury [48,54]. Preclinical studies showed that topically-applied AD-MSCs onto the exposed parietal cortex in middle cerebral artery occlusion (MCAO) rodent models had neuro-modulatory abilities by activating microglia and promoting astrocytosis and cellular proliferation within 3 days, which was associated with an improved neurological recovery [55]. A scaffold-free cell sheet allows implantation of a large number of AD-MSCs in the brain, which promotes angiogenesis, neurogenesis, anastomosis with the preexisting neurovascular unit, and behavior improvement in MCAO-induced rat stroke models [56].
AD-MSCs are also an important exosome source. AD-MSCsecreted exosomes, a type of extracellular vesicle containing protein, DNA, and RNA, can decrease ischemia-induced neuronal cell death [7]. Evidence shows that increasing miR-126 levels in the exosomes derived from AD-MSCs markedly improves functional recovery, promotes neurogenesis, and inhibits inflammation in stroke [7]. The importance of exosomes in stroke recovery was also explored in an in vitro study that identified the importance of miR-181b-5p. It plays an important role in regulating its target molecule, transient receptor potential melastatin 7, which promotes mobility and angiogenesis of brain microvascular endothelial cells after oxygen-glucose deprivation exposure (OGD) [57]. Therefore, AD-MSCs are promising as an exogenous exosome delivery system for ischemic brain injury treatment.
Bone marrow-derived MSCs
IV injection of BM-MSCs, but not AD-MSCs, improved survival rates, anti-inflammatory cytokine levels, and growth factors in a neonatal hypoxic-ischemic brain injury rat model [52]. BM-MSC administration protected the brain against ischemic injury after cardiac arrest and stroke by reducing inflammation, inhibiting the C-Jun N-terminal kinase pathway, and releasing exosomes containing miR-138-5p [22,58-60]. A translational phase I study of chronic stroke patients demonstrated the safety of intravenously transfused allogeneic, ischemia-tolerant BM-MSCs, as well as behavioral gains 1 year after treatment. This early study raised exciting potential for the application of this therapy in stroke [9]. Allogenic or autologous BM-MSC administration to stroke patients improved behavioral and functional outcomes [4,9,61].
Umbilical cord blood stem cells
Human umbilical cord blood stem cells (UC-BSCs) are derived from placental tissues, following birth. They consist of hematopoietic stem cells and MSCs. These cells offer a number of key advantages, such as an ample source of cells, low donor age, and low risks to babies and mothers during harvesting, which minimizes ethical concerns. These cells can differentiate into neural progenitor cells and provide neuroprotective effects in cerebral ischemia via neurotrophic factor secretion and vascular remodeling enhancement after stroke [62,63]. UC-BSCs have protective effects against ischemic injury, resulting in brainderived neurotrophic factor expression recovery [64]. In addition, UC-BSCs can inhibit the immune response and decrease the size of the ischemic brain lesion [65]. These results indicate their potential in ischemic brain injury.
However, these cells have a key disadvantage: they do not cross the blood-brain barrier (BBB) [66]. Interestingly, it was also noted that patients who underwent UC-BSCs transplantation were more prone to CNS infections [67].
Stem cell administration routes
Following the decision regarding the stem cell source, the most suitable therapy delivery method must be defined, which is complicated by factors such as sufficient BBB traversal, diffusion through tissues, uptake speed, and specific targeting.
Intravenous delivery
IV delivery circumvents the need to access the CNS directly, thus allowing for less invasiveness [60,68]. Intravenously injected NSCs can traverse the BBB and localize to ischemic injury areas [39]. NSCs, BM-MSCs, AD-MSCs, or UC-BSCs IV administration has shown neuroprotection in ischemic stroke [39,48,60,64,65,69].
Despite these results, stem cells delivered by the IV route are subjected to trapping in peripheral tissues (lungs, liver, spleen, and kidneys), particularly the lungs [70], limiting cell delivery to the brain. Additionally, pulmonary embolism complications were noted in a Korean family following IV AD-MSCs administration [71]. Cell size remains a major limiting factor in the intravenously delivered stem cell ability to traverse microvasculature or capillaries and avoid peripheral aggregation. Smaller cells, such as BM-MSCs (7 μm diameter), display up to a 30-fold greater passing rate through the lungs compared to larger stem cell types, such as AD-MSCs (18 μm) and NSCs (16 μm) [70,72]. Overall, it has been demonstrated that only a small number of cells reach the arterial system and target tissues, and that cells that get trapped in lungs can cause tissue damage [73]. In another study, IV BM-MSCs improved neurological function and restored BBB following damage from intracerebral hemorrhage [74]. However, BM-MSCs preferentially migrated to the spleen, being detected at a modest level at 12 hours and 11 days following treatment [75]. IV delivery of BM-MSCs in chronic stroke patients demonstrated an adequate safety profile [76]. UC-BSCs tail vein injection also reduced infarct area and restored bladder function that had been impaired by cerebral ischemia in a rat stroke model [69,77].
Intra-arterial delivery
Intra-arterial (IA) delivery uses catheterization to guide stem cells into the carotid artery or circle of Willis, thus bypassing the initial uptake by systemic organs. IA administration has been shown to be advantageous for MSC delivery at the site of injury [78-80]. This delivery method allows for bypass of the lungs and avoids pulmonary entrapment [72]. While this method delivers a large number of cells to brain lesions, it also carries a risk of micro-embolism and cerebral ischemia due to arterial cell aggregation and clumping, resulting in reduced cerebral blood flow [81-83]. In clinical applications, injection of BM-MSCs, both IA (internal carotid artery) and IV, demonstrated safety and showed improved functional scores at 4 to 8 weeks post-therapy in chronic stroke patients [76].
Intraventricular delivery
Intraventricular delivery allows for direct delivery into the cerebral spinal fluid (CSF) or interstitial tissue, thereby allowing the dispersal to multiple sites of the brain and CNS through the CSF. Despite the invasiveness of this technique, complications can be reduced with the proper training and technique. Intraventricularly injected BM-MSCs were transplanted into the lateral ventricles of cuprizone mice models, thereby initiating functional remyelination, oligodendrogenesis, and endogenous NSC proliferation through paracrine effects [84]. Intrathecally-administered UC-BSCs could migrate to the ischemic area and differentiate into neurons and astrocytes, which improved motor function in stroke rats [85]. Clinical applications of this method have supported the safety and potential efficacy, with improved neurological outcomes after intraventricular transplantation of BM-MSCs into patients with chronic sequelae of hemorrhagic stroke [86,87]. In cardiac arrest-induced global brain ischemia, human NSC infusion into the lateral ventricle 3 hours following resuscitation significantly improved neurological and electrophysiological outcomes of rats [88].
Intraperitoneal
Intraperitoneal (IP) administration of human MSCs (hMSCs) led to the rapid aggregation of cells in the peritoneal cavity, and only a small amount of cells migrated elsewhere [89]. When compared to hMSC IV injection, IP administration resulted in a lower accumulation in peripheral tissues, such as the lungs and liver. However, IP-delivered umbilical cord-derived mesenchymal stromal cells (UC-MSCs) showed poor localization to the ischemic frontal cortex [90], suggesting limited brain penetration.
Intraparenchymal
Localized intraparenchymal delivery allows for a higher cellular volume to reach brain target lesions [91], thereby preventing offtargeting effects and improving cost-effectiveness. Intracranial transplantation of human AD-MSCs was shown to promote neuronal repair in cerebral ischemia-reperfusion injury [54]. MSC intracranial injection has been used for neonatal hypoxic-ischemic brain injury [92]. iPSC subdural injection upon infarcted brain tissue can reduce the infarct area size and improve motor function after ischemic stroke [93]. Additionally, iPSC-derived NSC intracerebral transplantation was shown to differentiate into neurons and oligodendrocytes and improve recovery in a pig stroke model [94]. UC-BSCs injected into the contralateral cortex could also migrate to the ischemic region of stroke rats [95]. Despite these accomplishments, it remains controversial whether intracranial injections are feasible for clinical therapy [54,92,96]. Direct injection is invasive, as it requires a craniotomy, and this method can cause localized trauma and BBB damage [97]. The required surgery for this delivery method can potentially disrupt local niches and result in additional traumatic injury and inflammation. Intraparenchymal delivery is also ineffective at addressing multifocal disorders; thus, its feasibility for clinical translation is still debatable [96].
Intranasal delivery
Intranasal administration is a noninvasive and relatively effective drug or cell delivery route when compared to invasive intracranial transplantation. Although it has been recognized as a small agent (e.g., small molecules, proteins, viruses, bacteria, and nanoparticles) delivery method, its utility for cellular delivery has only been discovered in the last decade [98]. Intranasallydelivered cells collect beneath the nasal mucosa, close to the turbinate bones, and travel through the cribriform plate [99]. This method of delivery is non-invasive, rapid, and offers BBB bypass [100]. Cells showed improved targeting and less accumulation in peripheral organs, compared to systemic dosage [101]. Additionally, intranasal delivery allows for repeated dosages [102-105], making it more practical than other methods.
Both MSCs and NSCs have been shown to be effective agents in intranasal cell therapy [102,106]. Intranasal delivery of hypoxiapreconditioned BM-MSCs in a rat stroke model demonstrated significantly reduced cell death in peri-infarct regions [107]. Intranasal MSC treatment of neonatal hypoxic ischemic-injured mice has demonstrated long-term cognitive and sensorimotor improvement [105]. Furthermore, a single intranasal dose was sufficient for therapeutic benefits, while at least two intracranial dosages were required to achieve the same effect. Intranasallyadministered NSCs to HIE neonatal rats showed substantial migration into the brain 24 hours post-delivery and improved functional neurological outcomes [108]. In stroke, BM-MSC intranasal delivery provided neuroprotection, enhanced neurovascular regeneration, and led to improved functional recovery [63,103,109].
It is important to recognize that the clinical translation of this method remains poorly understood. Humans have a smaller olfactory bulb compared to that in the rodents utilized in these preclinical studies, and it remains unclear if this difference can hinder the translational potential of intranasal administration. Of note, a hypoxic-ischemic brain injury neonatal primate model demonstrated migration of intranasally delivered MSCs to the site of injury, suggesting a feasible translation [96].
Overall (Table 1) [42,45,51,77,78,81,82,84,90,91,100,102-105,110], IV, intracerebral, and IA routes have been the most utilized and investigated for stem cell administration in preclinical and clinical studies. IV administration facilitates widespread implementation and avoids invasive procedures, but reduces brain tissue penetration due to cell diffusion and risks accumulation in other organs such as the liver, spleen, and lungs [111]. Intracerebral injection has high therapeutic benefits, but with invasive risk. Conversely, the IA route has better CNS penetration and bypasses other organ systems, but has associated arterial micro-embolism and cell-aggregation risks [111]. Intranasal delivery, a minimally invasive novel delivery mechanism, may be an optimal route in the future, but technical aspects, such as administration technique, cell type, position of head during administration, and volume of dosage should all be considered, requiring further investigation in clinical application.
Improvement of stem cell application in ischemic brain injury
Multiple factors influence cell transplantation efficacy and treatment outcomes. Ideally, stem cells used in the treatment of CNS disorders need to be highly manipulated, to be integrated into the host tissue, and should be able to traverse the BBB [112-114]. They must overcome the limitations of scarce donor sources, uncontrolled cell maturity, poor cell survival, inefficient delivery, and low engraftment rate [115] prior to their utilization as a viable clinical therapy. A major difficulty within stem cell therapy is the inability to maintain cell viability, properties, and functions before and after in vivo implantation. This can lead to low cell survival rates and poor outcomes in growth, homing, differentiation, and paracrine effects [116]. To overcome these obstacles, methods such as stem cell modification, pretreatment, and chemical or biomaterial assistance, have been used to enhance stem cell therapy and promote its clinical application.
Optimal dose, timing, and age of stem cells
Two essential parameters that can be optimized in the use of stem cells in ischemic brain injury include the number of delivered cells and the timing of delivery. A density ranging from 5×103 to 2×107 MSCs has been studied in animals, typically intravenously, immediately following ischemic stroke or over a period typically varying from 24 hours to 30 days [111]. Preclinical studies indicate that IV doses ranging from 3.6×104 to 4.3×107 MSCs/kg body weight provide substantial behavioral gains in rodents [117]. An additional study noted a significant lesion size reduction and sensorimotor function improvement when 0.5×106 MSCs were delivered per mouse in a neonatal hypoxicischemic model. They noted that decreasing the dose to 0.25×106 MSCs did not have a significant effect on sensorimotor performance or gray/white matter preservation, while increasing the dose to 1.0×106 did not result in additional improvements to sensorimotor function or lesion size reduction [105]. Translationally, IV transfusion of 0.5, 1.0, and 1.5×106 allogeneic BM-MSCs/kg body weight was shown to be safe in a phase I trial and suggested behavior gains in chronic stroke patients [9]. UC-BSCs (2×106 cells per rat) showed safety and neuroprotection in stroke rats after IV administration 2 days following ischemia induction [77]. Additionally, IV injection of UCBSCs (1×106 or 4×106), 1 day after ischemic injury, has been shown to reduce neurological injury in stroke rats [118].
Regarding timing, MSCs administered 3 or 10 days postneonatal hypoxic ischemic injury showed significant improvements in motor behavior, while administration at 17 days showed neither sensorimotor function improvement nor gray/white matter loss reduction. This suggests a broader treatment window for intranasal MSC delivery, particularly when compared to the current treatment window of 6 hours for therapeutic hypothermia, which is the current standard of care [105]. In another study, hypoxic-preconditioned BM-MSCs were intranasally delivered 3, 4, 5, and 6 days after ischemic stroke in mice. These cells reached the brain 6 hours after delivery, where they increased neurogenesis, angiogenesis, and local cerebral blood flow when measured 21 days after stroke. Functional recovery was improved in these animals. This study highlighted that repeated administration of stem cells 3 to 6 days after stroke could be beneficial, thus extending the therapeutic window for stroke beyond the current 24-hour limitations [103]. BM-MSCs administration between 2 and 7 days of injury significantly increased health benefits compared to 12 to 24 hours and >7 days [119]. Delivery at 7 days post-stroke has been most commonly reported in the literature, which may be related to the specific brain environment at that time, with decreased glutamate and brain edema [120]. After stroke, the brain microenvironment is constantly changing, with increased excitotoxicity, inflammatory reaction, and trophic factor expressions [120]. Immediately following ischemic insult, ischemia-reperfusion events occur, including cellular energetic failure, mitochondrial Ca2+ overload, oxidative stress, BBB dysfunction, inflammation, apoptosis, and eventual neuron and glial cell necrosis [121,122], which may block or interfere with the transplanted stem cells’ neuroprotective effects. Thus, the pro-regeneration environment in the ischemic area around the 7-day time point may be considered as the optimal time point for stem cell delivery. Compared with ischemic stroke, brain injury induced secondary to cardiac arrest has different conditions related to stem cell treatment (Table 2) [10,16-18,51,123-127]. To date, BM- and AD-MSCs are the most commonly used stem cells in this pathology, and the cells are typically administered 1 to 2 hours after the return of spontaneous circulation. After 1 hour of brain ischemia, the subdural transplantation of iPSCs (1.0×106) is found to improve recovery in stroke rats [93]. Recently, our lab reported that intraventricularly administered hNSCs 3 hours after 8-minute asphyxia-cardiac arrest significantly improved neurological outcomes [88]. However, an optimal stem cell delivery timing has not been fully elucidated regarding this pathology, and additional studies are needed to provide further evidence for optimal time point of stem cell delivery and timedependent pathophysiologic changes of the brain after cardiac arrest. Available preclinical studies indicate that IV doses ranging from 1×106 to 5×106 cells provide substantial behavioral gains in post-cardiac arrest rodents [10,16-18,51,123-125]. According to the allometric scaling approach from animals to humans recommended by the Food and Drug Administration, the equivalent MSC dose in humans is about 1.6×106 MSCs/kg [9].
The hMSC donor age is another critical factor affecting functional and structural outcomes in stroke affecting functional and structural outcomes in stroke [128,129]. MSCs from older donors have a decreased proliferative capacity and trophic factor secretion [130]. Autologous transplantation has been suggested as a safe therapy with minimal immune consequences. However, stroke patients tend to be older, which limits the proliferation potential of their autologous stem cells and limits this mode of therapy in this particular pathology.
Stem cell dosage and timing remain to be optimized, both to avoid complications, such as vessel embolism and local energy deficiency, and to optimize transplanted cell survival, differentiation, and function. To achieve this, stem cell interactions with the dynamic microenvironment in the ischemic brain must be further understood. Stem cell therapy should be considered according to the delivery routes, stem cell types, and quality, progress of disease, and age of patients for optimal translation in dose, timing, and cell age.
Stem cell preconditioning
Hypoxic preconditioning
Stem cell hypoxic preconditioning has been shown to increase cell survival and promote angiogenesis and neurogenesis [131]. It also improves MSCs homing and neuroprotective ability [79,98]. Under low-oxygen (5%) culture conditions, MSCs have a higher proliferation rate, cytokine secretion, and migration activity [132,133]. Hypoxic-preconditioning of BM-MSCs enhances cell homing and increases regeneration and functional recovery after ischemic stroke [75,107]. In vitro, hypoxia conditioning improves the protective efficacy of aged human BM-MSCs in OGD-induced neuronal injury by increasing the secretion of vascular endothelial growth factor (VEGF) [15]. Hypoxia-preconditioned MSCs in vivo transplantation promotes MSC migration and integration and decreases neuronal death in cardiac arrest-induced ischemic brain injury [126].
Bioactive factor overexpression
Enhancing the expression of specific trophic factors is an important method for stem cell modification. Mechanistic analysis suggests that MSCs’ therapeutic efficacy against neurologic disorders may be mediated by VEGF and brain derived neurotrophic factor (BDNF) secretion [134,135]. Further evidence shows that both BDNF and VEGF overexpression in BM-MSCs markedly enhances neuroprotective potency from cardiac arrest-induced global cerebral ischemia one week after cell transplantation [17]. Increasing the expression of glial cell line-derived neurotrophic factor (GDNF) in UC-BSCs markedly elevates GDNF level in the infarcted hemisphere and enhances stroke rats’ functional recovery [136].
Fibroblast growth factor type 1 (FGF1) is abundant in sensory and motor neurons and binds to FGF receptors, which contributes to neuroprotection mediated by autocrine, paracrine, and intracrine pathways [137,138]. In vitro FGF1 (100 ng/mL) exposure can promote neurite outgrowth of cortical neurons, and application of FGF1 (1 or 2 μg) in the brain injured area improves neuroprotective effects in a cerebral ischemic rat model [139]. Acute IV administration of 2×106 FGF1-overexpressed ADMSCs was shown to improve functional neurological recovery and mildly reduce infarct volume in a stroke rat model [140].
A novel optochemogenetics fusion protein, luminopsin 3, can be activated by either extrinsic physical light (i.e., laser and light-emitting diode) or by the luciferase substrate coelenterazine. Stimulation of luminopsin 3 and iPSC-derived neural progenitor cells increases synapsin-1, postsynaptic density 95, BDNF, and stromal cell-derived factor 1 expression. Focal ischemic stroke mice receiving the combined luminopsin 3-iPSCneural progenitor cells (NPCs)/coelenterazine treatment possess enhanced neural network connections in the infarct region, thereby promoting optimal functional recoveries [14].
Chemical treatment
Previous reports indicate that metformin is an optimal neuroregenerative agent, acting on multiple stages, such as proliferation (dependent on the p53 family member and transcription factor TAp73), differentiation (activating the AMP-activated protein kinase [AMPK]-atypical protein kinase C [aPKC]-CREBbinding protein [CBP] pathway), and cell survival [141,142]. Metformin, a known AMPK activator, has neuroprotective effects for stroke treatment and prevention. IP injection of metformin (50 mg/kg) prior to stroke for 3 weeks improved stroke-induced lactate generation and ameliorated mouse stroke injury [143]. Moreover, rats preconditioned with metformin (10 mg/kg, IP), prior to inducing ischemia, had reduced apoptotic cells in the peri-infarct region [144]. Metformin (50 μM)-preconditioned iPSCNSCs promote post-stroke recovery by enhancing hiPSC-NSC proliferation, differentiation, and engraftment [145].
Minocycline, a semisynthetic tetracycline, also has a neuroprotective potential in cerebral ischemia. Treatment with minocycline (45 mg/kg IP twice a day for the first day; 22.5 mg/kg for the subsequent 2 days) before or after ischemia has been shown to reduce the infarct size in the cerebral cortex, and minocycline therapy has a wide therapeutic window [146]. Minocycline treatment (10 μM) increases NSC proliferation capacity, and minocycline preconditioning induces NSCs to release paracrine factors, including BDNF, GDNF, nerve growth factor, and VEGF [147]. In addition to the anti-inflammatory effects in ischemic brain injury [148], minocycline preconditioning can also improve NSC survival after exposure to ischemic reperfusion injury via nuclear factor erythroid 2-related factor 2 (Nrf2) and heme oxygenase-1 upregulation [147].
CD34+ UC-BSCs pre-treated with 17β-estradiol were shown to reduce the infarct size at 2 hours and 2 days in a rat stroke model. This pretreatment also improved cerebral blood flow at 2 hours and reduced hyperperfusion at 2 days. This study was performed in female ovariectomized rats with a proposed mechanism of stabilization of cerebral hemodynamics and BDNF expression regulation [64].
Others
Short-term exposure to oxidative stress at low concentrations has been shown to improve NPC resistance to acute stress [149]. Three-dimensional (3D) stem cell aggregates can also precondition cells by increasing extracellular matrix secretion and promoting trophic functions [131]. Electrically preconditioned NPCs with a conductive polymer scaffold improved functional outcomes compared to unstimulated cells in stroke [150]. Stroke patients' serum from the acute phase of stroke can activate MSCs, and markedly promote neurogenesis and angiogenesis [151]. Ultimately, preconditioning has been shown to result in a suppressed immune response, increased regeneration, enhanced migration and paracrine effects, and increased treatment efficacy [152].
Overall, to enhance stem cell-mediated neuroprotection in stroke, preconditioning of stem cells with hypoxia, electrical stimulation, chemicals, or neurotrophic factor overexpression might be an important strategy for their future clinical application (Figure 1). These preconditioning methods were majorly based on mechanisms of stem cell therapy in stroke. Thus, it is significant to clarify the molecular modulation of stem cells in neuroprotection, which may lead to the discovery of more preconditioning methods.
Combination approaches
In cardiac arrest-induced brain injury, MSC-treated rats exhibit better or equivalent functional outcomes, compared to rats treated immediately with therapeutic hypothermia (32°C to 34°C) for 2 hours after ischemia, although hypothermia was not found to markedly enhance MSC therapy benefits in this pathology [51]. However, in ischemic stroke rats, post-ischemia hypothermia induced by injection of cold saline (4°C) via carotid artery at an infusion rate of 0.6 mL/min for 5 minutes can enhance the therapeutic effects of MSC on reperfusion injury [153].
Seven days after ischemia induction, the combined utilization of UC-BSCs (injection once via the tail vein) and erythropoietin (IP exposure for 5 consecutive days) was shown to have strong neurogenic and angiogenic effects in stroke rats [154]. Fibrin glue, a biocompatible and biodegradable natural product, combined with iPSCs enhanced stoke-induced deficits recovery. The fibrin glue may also increase iPSCs utilization safety [93].
Cerium oxide nanoparticles can bind to oxygen molecules and may scavenge intracellular reactive oxygen species [155]. Hyaluronic acid-cerium oxide-labeled human umbilical cord MSCs gain combined antioxidant and anti-inflammatory effects, and significantly attenuate stroke injury [156].
Enhancement of stem cell delivery in brain
Increasing BBB permeability
Mannitol, a hyperosmolar extracellular agent often used to control intracranial pressure following brain injury, may serve as a useful agent to increase BBB permeability. Coupling intravenously delivered UC-BSCs with mannitol to facilitate the entry of therapeutic biologics into the brain showed improved neuroprotection when compared with UC-BSCs alone [66]. The combination drug treatment of mannitol and temozolomide increases BBB permeability through the inhibition of endothelial tight junction proteins, and allows for the efficient delivery of UC-MSCs microvesicles into the brain in a chronic stroke rat model [157]. Focused ultrasound, in combination with magnetic fields, can also enhance stem cell delivery by selectively opening the BBB at specific predetermined locations [158].
Biomaterials
Biomaterials systems continue to play an increasingly important role in regenerative medicine, particularly alongside stem cell usage. They can be used to form scaffolds that encapsulate growth factors and cells in order to protect and shield them from unfavorable in vivo environments, while sequestering factors to promote an optimal regenerative environment [159]. Encapsulation also prevents in vivo immune response activation and inflammation [131]. Additionally, these scaffolds may intrinsically contain properties that can provide cues to stem cells such as matrix topology, mechanical forces, and biochemical properties [160]. For example, various studies have demonstrated that softer substrates can promote neurogenic differentiation in NSCs, while stiffer substrates will instead direct them towards astrocyte differentiation [161]. This highlights the need to optimize these biomaterials for stem cell viability, differentiation, and proliferation. A recent meta-analysis found that biomaterialbased interventions have led to lesion volume reduction and improved neurological outcomes in stroke rodent models [162].
Hydrogels act as 3D scaffolds that improve stem cell survival and differentiation, both in vivo and in vitro [163]. However, they must be finely engineered for optimal cell delivery. Gels that solidify too quickly can get clogged in a needle during delivery. However, if they solidify too slowly, the therapeutic agent may diffuse out of target sites, thus resulting in suboptimal therapy [164]. Injectable chitosan self-healing hydrogels mixed with murine NSCs were synthesized to address this issue and showed promise for CNS injury. Chitosan is a nontoxic biodegradable substrate that can promote nerve regeneration. The authors found that neurosphere progenitors grew twice as fast in selfhealing hydrogels and had a greater tendency to differentiate into neuron-like cells [164]. Another study demonstrated that matrigel scaffolding plus neuronal precursor cell injection into the infarct cavity could improve the functional outcome, with notable effects several weeks after initial stroke injury [165]. Neuroepithelial progenitor cells encapsulated in a hyaluronan-based hydrogel showed facilitated injection of the cells into the brain and reduced inflammatory response in the stroke-injured rodent brain [163]. Injectable hydrogels composed of hyaluronan acid and methylcellulose were demonstrated to improve the survival and integration of stem cells following transplantation in stroke injury murine models [166]. Methylcellulose promotes neuron regeneration through axon connections, while hyaluronan acid promotes angiogenesis and cell proliferation [167]. The bioactive materials that compose this hydrogel provided survival factors that enhanced NSC distribution and survival in the brain via CD44-mediated mechanisms and inhibited apoptosis [168]. Injection of a biopolymer hydrogel, together with neural progenitor cells, into the infarct cavity after stroke was shown to significantly enhance the survival of transplanted cells and diminish inflammatory response [169]. Post-polymerization modification of methionine-based co-polypeptides demonstrate tunable properties that aid in vivo transplantation of neural stem/progenitor cells in the CNS. In the absence of growth factors, these hydrogels preserved the transplanted cells and facilitated local axon regrowth, retained transplanted cells close to the injection site, and allowed integration into the host neural tissue [170].
Stem cell magnetic retention in target location
Magnetic cell targeting can guide cells to sites of injury to overcome stem cell retention limitations after intra-carotid or IV delivery. Human neuro-progenitor cells labeled with ultrasmall superparamagnetic iron oxide nanoparticles were used to increase efficacy and retention at the site of traumatic brain injury (TBI) in rats. Labeled cells did not exhibit any modifications in viability, proliferation, or differentiation, thereby suggesting that neither nanoparticles nor magnetic fields are disruptive to the intrinsic behaviors of the cell [158]. This method of enhanced targeting remains to be applied to ischemic brain injury, but its efficacy in TBI shows promise for other brain pathologies (Figure 1).
In summary, stem cell therapeutic strategies include the enrichment of stem cells in the brain, stem cells engineered with biomaterials, and stem cell control with magnetism in an effort to overcome the weaknesses of stroke stem cell therapy. Stem cell engineering has facilitated an enhanced docking ability in the endothelium within the ischemic core. Magnetic retention of stem cells is promising to target the location of ischemic injury within the brain. Stem cell lineage differentiation induced by chemicals or biological factors may contribute to tissue regeneration in the stroke infarct bed. More methods, such as the combination of various stem cells, biomaterials, genetic, and chemical modifications, may emerge in future studies to enhance stem cell therapy in stroke and global cerebral ischemia.
Stem cell tracking
To track the transplanted cells, stem cells are labeled with probes such as fluorophobes (4′,6-diamidino-2-phenylindole [DAPI], PKH26 [red fluorescent dye], or 5-bromo-2´-deoxyuridine [BrdU]) [16,125,126], radiotracers, or paramagnetic nanoparticles before transplantation, and are then monitored in vivo using optical imaging, single photon emission computed tomography (SPECT), positron emission tomography (PET), or magnetic resonance imaging (MRI) [171]. Two pitfalls of direct labeling include dilution of the labeling agent with proliferation of originally-labeled cells and in vivo uptake of probes by phagocytic cells. Indirect methods of tracking consist of exogenous reporter genes that have been delivered to the cells of interest, such as ferritin heavy chain, which can act as robust MRI reporters to track cell distribution and migration for acute ischemic stroke [172]. Since only surviving cells take up the reporter, viable cells can be clearly visualized. However, gene delivery may disrupt normal cell physiology [173].
Direct tracking via MRI provides advantages, such as morphology characterization, high spatial resolution, lack of radiation, and long-term stem cell monitoring [174,175]. MRI requires the use of a contrast agent to visualize cells, e.g., superparamagnetic iron-oxide nanoparticle (SPION), which has been shown to allow in vivo retention of neural progenitor cell viability, phenotype, proliferation, and differentiation [176,177]. Highspeed MRI has been used to visualize IA delivery of SPION-labeled stem cells in both control and stroke animal models. Real-time feedback allowed for accurate spatial and temporal distribution information. It also allowed cell infusion speed to be optimized in response to vascular resistance. This method also provided information on stem cell delivery efficacy, destinations and off-target routes, adherence, and aggregation [178]. Long-term MRI tracking of intraventricularly-delivered SPIONlabeled UC-BSCs has been demonstrated in a 9-month-old global cerebral ischemia patient. The patient was monitored over 33 months, during which the signal decreased over 4 months and became undetectable at 33 months [179].
Despite these promising results, MRI is limited by its need for iron-based visualizing agents. These agents can cause false-positive signals from microphages that have engulfed imaging agents, iron being deposited in the brain, or small hemorrhage [180]. Other limitations include poor quantification and sensitivity [171]. A dual-contrast method, which utilized slow-diffusing SPION and fast-diffusing gadolinium-based chelate-labeling properties, was used to feasibly detect cellular migration and death [175]. This may advance stem cell therapies by allowing the visualization of transplanted cells’ viability and analysis of their in vivo survival. Studies have demonstrated the use of MRI to track micron-sized paramagnetic iron oxide-labeled MSCs injected intranasally to confirm their migration towards glioma xenografts [102]. MnEtP, a manganese porphyrin contrast agent, also has an excellent sensitivity and specificity for non-invasive stem cell tracking detected by MRI [181]. MSCs labeled with superparamagnetic iron oxideloaded cationic polymersomes can also be tracked in vivo using MRI [182].
SPECT radiotracers, particularly indium 111 (111In) and technetium-99m (99mTc) as well as PET, are also promising. These methods provide sensitivity in the picomolar range and ability to use the same tracer across multiple species. However, they do not provide anatomical information and must be used in conjunction with MRI, computerized tomography, or X-ray. SPECT imaging has the added benefit of a lower false-positive signaling compared to MRI [27,183].
Near-infrared (NIR) fluorescence imaging is advantageous over conventional fluorescence imaging due to its improved ability to penetrate tissues. NIR fluorescence imaging was shown to visualize and track the in vivo tumor-targeting process and tissue distribution of intravenously injected MSCs [184]. This technology may allow the visualization and effective targeting of cells to ischemic tissues.
Clinical translation and future perspectives
The extensive preclinical literature base would suggest that stem cell therapies for the treatment of ischemic brain injuries are closer to becoming a realistic clinical option [185]. Several phase I clinical studies have confirmed the safety and feasibility of stem cell therapy in stroke, though efficacy and clinical practicality continue to require refinement [9,186].
The application of these findings in human studies has led to two approaches in clinical trials: acute administration to prevent ischemic injury secondary to stroke versus late administration in the chronic stroke phase with the goal of promoting neuronal regeneration. A recent phase I trial investigated IV administration of autologous bone marrow mononuclear cells within 24 to 72 hours of acute ischemic stroke onset, and patients were followed up for 2 years. No severe adverse effects were detected, thus highlighting the safety of these cells, but the study was not powered for functional outcome effects [13]. A second trial delivered allogenic BM-MSCs depleted of CD45/glycophorin-A within 48 hours of ischemic stroke and reported a non-significant trend toward improved functional outcomes in the treatment group with no severe adverse events [61]. In stark contrast to the wealth of preclinical supportive data for this therapy, the largest available phase II randomized controlled trial with blinded endpoint performed in five centers in India found no significant difference in 6-month clinical outcome of ischemic stroke after IV injection of autologous BM-MSCs to 60 acute ischemic stroke patients at a median of 18 days after stroke [4]. Other studies have investigated the chronic model for stem cell use in stroke management. An open-label pilot trial on 12 chronic stroke patients has demonstrated improved behavioral outcomes 208 weeks following transplant and no adverse immune effects after autologous BMMSCs transplantation [187], and a longitudinal trial following patients for 2 years post-NSC transplantation showed improved NIHSS scores and no adverse effects of treatment [45].
To date, the use of stem cells in the treatment of post-cardiac arrest global cerebral ischemia remains in the animal study phase of investigation and has shown improved neurological functional outcomes in multiple studies of rat cardiac arrest models. Proposed mechanisms include immune/inflammatory modulation, secretion of BDNFs, and BBB integrity protection [123,188]. The clinical use of therapeutic hypothermia is the only available treatment for neuroprotection following global cerebral ischemia induced by cardiac arrest [189]. Studies have demonstrated the use of MSCs in conjunction with therapeutic hypothermia after cardiac arrest, thereby reporting increased neuronal survival, BBB protection, reduced oxidative stress, and reduced neutrophilic infiltration [51]. Given the morbidity and mortality of global cerebral ischemia secondary to cardiac arrest and the paucity of management strategies to improve functional outcomes, MSC treatment should be escalated to larger primate or human models. There are currently no registered clinical trials in this area.
Approximately 55 clinical trials have been registered to investigate the effects of multiple stem cell types, including MSCs derived from the adipose tissue, bone marrow, and umbilical cord, NSCs, hematopoietic stem cells, endothelial progenitor cells, placenta stem cells, multilineage differentiating stress enduring (muse) cells, and modified stem cells on ischemic stroke treatment (https://www.clinicaltrials.gov/) (Figure 2A and B). Owing to weak support in the clinical data-to-date, studies investigating the optimal timing, dosing, and cellular modifications for improved functional outcomes and reduced infarct burden are imperative. Currently, 19 clinical trials have entered into phase 2, and Figure 2C summarizes trials that are now under way.

Stem cell therapy clinical trials in ischemic brain injury. (A) Number of clinical trials in the world, (B) according to the stem cell types. Other stem cells including placenta-derived cell, endothelial progenitor cell, hematopoiesis stem cell, muse cells, and modified stem cells, and (C) according to the recruitment status (clinicaltrials.gov, March 2020). UC-MSC, umbilical cord-derived mesenchymal stem cell; PSC, placenta-derived cell; UC-BSC, umbilical cord blood stem cell; SC, stem cell; NSC, neural stem cell; EPC, endothelial progenitor cell; HSC, hematopoietic stem cell; BMSC, bone marrow stem cell; AD-MSC, adipose-derived mesenchymal stem cell.
Our review suggests that utilization of novel delivery techniques, particularly intranasal delivery, should garner special attention in the application of stem cell therapy to ischemic brain injury patients and escalation to primate and human studies is warranted. The use of encapsulated scaffolds and cellular preconditioning may also offer advancement in the application of stem cell therapy for these patients (Figure 1).
Conclusions
Studies continue to optimize stem cell therapies and address the limitations, including the ability to effectively target the CNS, remain therapeutically viable in vivo, and exert beneficial effects while simultaneously avoiding adverse consequences. Novel techniques, such as intranasal delivery, seem promising and need to be validated in adult primates or humans to fully elucidate their potential for stem cell-based therapies. To further advance this field, methods that combine optimal delivery, dosages, preconditioning, and tracking will be needed and should be explored in varying cerebral ischemia models.
Notes
Disclosure
The authors have no financial conflicts of interest.
Acknowledgements
The work was partially supported by R01HL118084 and R01NS110387 from the National Institutes of Health (NIH) (both to Xiaofeng Jia).