Introduction
Unruptured intracranial aneurysms are not uncommon. The reported prevalence in the general population is 1%-9% in autopsy series1,2,3,4 and 0.5%-2% in imaging studies.4,5 This wide range in reported prevalence may be attributable to variations in evaluation methods and demographics. The diagnostic accuracy for unruptured intracranial aneurysms has increased6 following the recent advent of non-invasive vascular imaging tools such as multi-channel computed tomography angiography and magnetic resonance angiography (MRA), and incidental asymptomatic aneurysms have come to represent a substantial clinical burden on account of their controversial natural history.
At our institution, a head MRA is performed for not only patients with ischemic stroke or a high number of cerebrovascular risk factors but also preoperative evaluation of patients with a planned major cardiac or transplantation surgery. Head MRA has also recently become a component of routine health check-ups in selected cases when concerns regarding cerebrovascular disease are present. On several occasions, we were able to identify various cerebrovascular lesions such as steno-occlusive lesions. We further noted a gradual increase in the incidence of incidental asymptomatic aneurysms. Therefore, the aim of this study was to describe the incidental intracranial saccular aneurysms detected on MRA performed at our institution over a 5-year period. We report the prevalence, anatomical location, size, and clinical follow-up findings of intracranial saccular aneurysms, as well as the gender and age of patients with incidental intracranial saccular aneurysms.
Methods
Patient selection
A search of the radiology report database identified 19,171 sets of MRA examinations from 18,237 patients that were obtained between January 2001 and December 2005. This period was selected so that at least 5 years of follow-up data could be available for all patients. All patients were ethnic Koreans. MRA was performed to detect various cerebrovascular diseases, to evaluate nonspecific cerebrovascular symptoms such as dizziness or dementia, as part of a standard health check-up, or as part of a preoperative evaluation in patients with planned major cardiac or transplantation surgery.
Patients with incidentally detected aneurysms were identified from the radiology reports (Figure 1). Patients with known intracranial aneurysms prior to the MRA examination and those with clear dissecting aneurysms or fusiform aneurysms were excluded. Patients with subarachnoid hemorrhage detected at the time of magnetic resonance (MR) imaging were also excluded. Patient data including age, gender, symptoms, clinical treatment course, and follow-up results were obtained from medical records.
Acquisition of MR imaging sequences
MR images were acquired on one of six 1.5-T MR scanners (2 Siemens Magnetom Avanto, Erlangen, Germany; 1 GE Signa CV/i, Milwaukee, USA; and 3 Philips Gyroscan Intera, Best, The Netherlands). A ramped radiofrequency pulse and three-directional zero filling were used during data acquisition. MRA data were acquired using the three-dimensional time-of-flight method focusing on the circle of Willis with the following parameters: echo time, 7 ms; repetition time, 25 ms; flip angle, 20°; one excitation; field of view, 160 or 200 mm; matrix size, 256×512; voxel size, approximately 0.9×0.9×1.0 mm; slab number, 4. The first set of 12 maximum intensity projection images was generated by rotating around the sagittal axis at 15°. For the second and third sets of maximum intensity projection images, sub-volumes that contained the right and left circulation, respectively, were selected and 12 maximum intensity projection images were generated for each by rotating around the sagittal axis at 15° to construct 24 images. In total, 36 images providing 12 stereoscopic images in three orthogonal directions were examined on the picture archiving and communication system for each patient. If more images were required due to the delicate delineation of the arterial structures, random image projections of volume rendering were created by reconstructing the source images using an Advantage Workstation (GE, Milwaukee, WI, USA).
Image interpretation
"Aneurysm" was used as a wild-card search word to identify the initial candidate cases of incidental aneurysms from among the 19,171 sets of MRA examinations contained in the radiology report database. Two experienced neuroradiologists (C-WR and HWP) used MRA images and medical reports to confirm the presence of an aneurysm and to quantify the size and location of the aneurysm. If there was a discrepancy between the original report and the reader's interpretation, or if the presence of an aneurysm was equivocal, another neuroradiologist (DHL) reviewed the records in order to reach a consensus. False-positive cases, in which the radiology report appeared in the initial search results but no aneurysm was found upon re-examination of the MRA images and medical records, were recorded and the cause of the false-positive was noted.
Aneurysm size was defined as the largest diagonal measurement seen on the maximum intensity projection images and classified as one of four categories modified from a previous study:7 <2 mm, 2-7 mm, 7-12 mm, and 13-24 mm. Aneurysm location was described according to the secondary artery arising from the parent artery and classified as: paraclinoid internal carotid artery (ICA), distal ICA posterior wall, ICA bifurcation, anterior communicating artery, anterior cerebral artery, middle cerebral artery (MCA) trunk, MCA bifurcation, distal vertebral artery, basilar trunk, basilar top, posterior cerebral artery, superior cerebellar artery, or posterior inferior cerebellar artery. Paraclinoid ICA aneurysms included aneurysms originating near the anterior genu of the cavernous ICA or near the ophthalmic artery, and distal ICA posterior wall aneurysms included ICA aneurysms originating at the posterior communicating artery or the anterior choroidal artery. Multiplicity was checked and aneurysm numbers were recorded. In patients with multiple aneurysms, one index aneurysm with the largest size was selected for prevalence calculations. Patient age was categorized as 20-29 years, 30-39 years, 40-59 years, 60-79 years, 70-79 years, or ≥80 years. Paired t-tests were performed to evaluate statistical differences in prevalence according to overall gender. Linear regression analyses were performed to test the relation between prevalence and age in men and women.
Results
Prevalence of incidental aneurysms
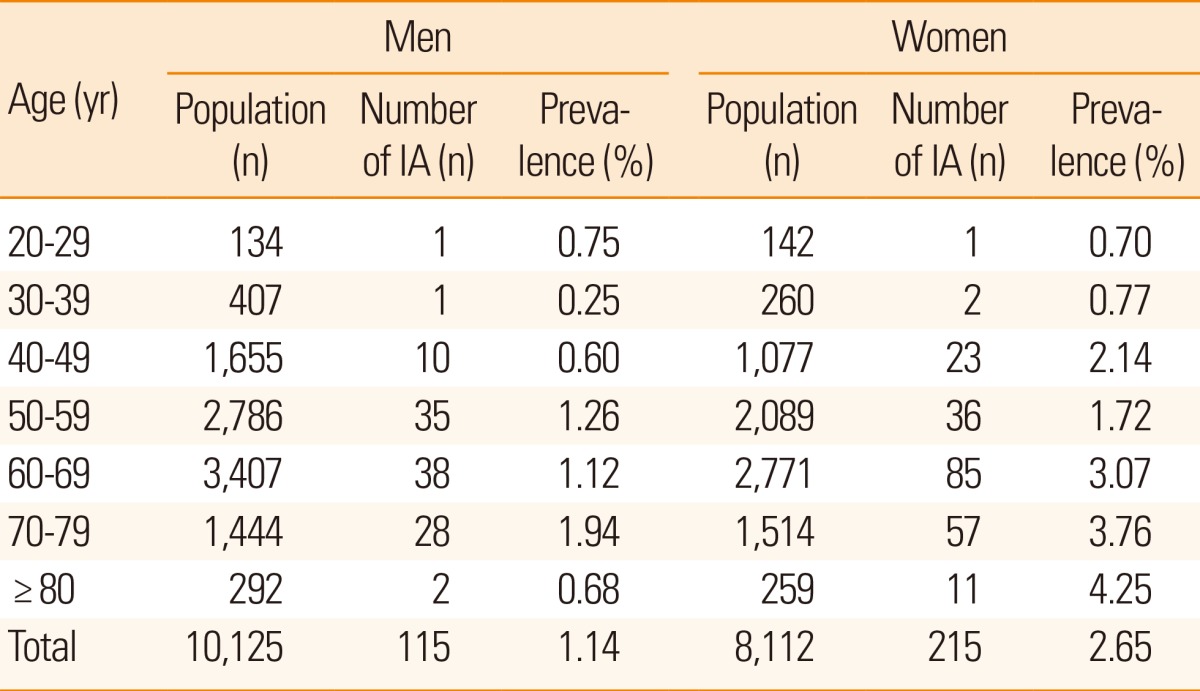
Of the 18,237 patients studied, 330 (115 men and 215 women) had incidental aneurysms, resulting in a prevalence of 1.8% (95% confidence interval, 1.6%-2.0%). The median age of patients with incidental aneurysms was 63 years and the age range was 22-82 years. Multiple aneurysms were found in 30 patients (9.1%), including 26 patients with 2 aneurysms each, 3 patients with 3 aneurysms each, and 1 patient with 4 aneurysms. A total of 366 aneurysms were identified in 330 patients. Of these patients with incidental aneurysms, those assessed as part of a health check-up (n=165) had more frequent aneurysms than patients with a previous stroke history (n=136) or cardiac risk (n=29).
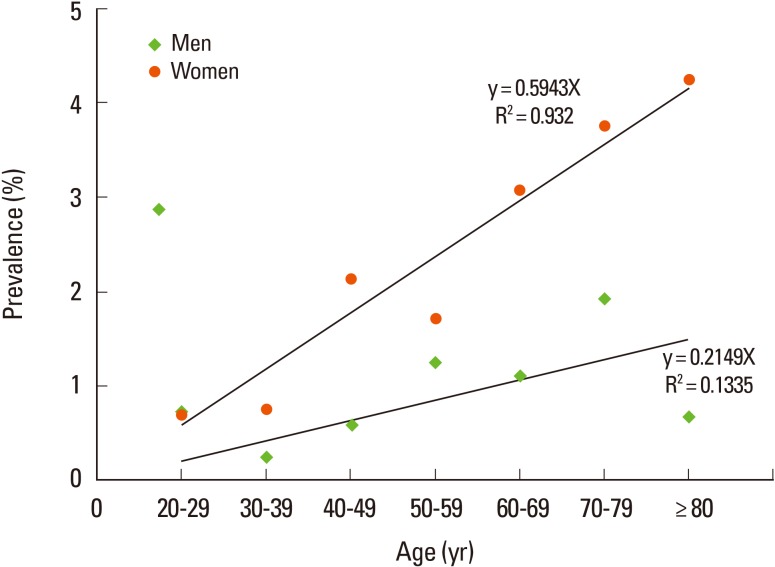
The prevalence of incidental aneurysms was significantly higher in women (215/8,112) than in men (115/10,125; P=0.02; Table 1; Figure 2) and nonsignificantly higher in men aged 70-79 years (28/1,444) than in men aged >80 years (2/292; P= 0.30; Table 1; Figure 2). Among women, the prevalence of aneurysms was highest in women older than 80 years in a linear regression analysis (P<0.01). Prevalence increased with age in women (P<0.01), but not in men (P=0.30).
Size and location of incidental aneurysms
Aneurysm size ranged from 1.5 mm to 13 mm, and the median size was 4.0 mm. Most aneurysms (299/344; 86.9%) were in the 2-7 mm category. Only two aneurysms measured <2 mm (0.6%).
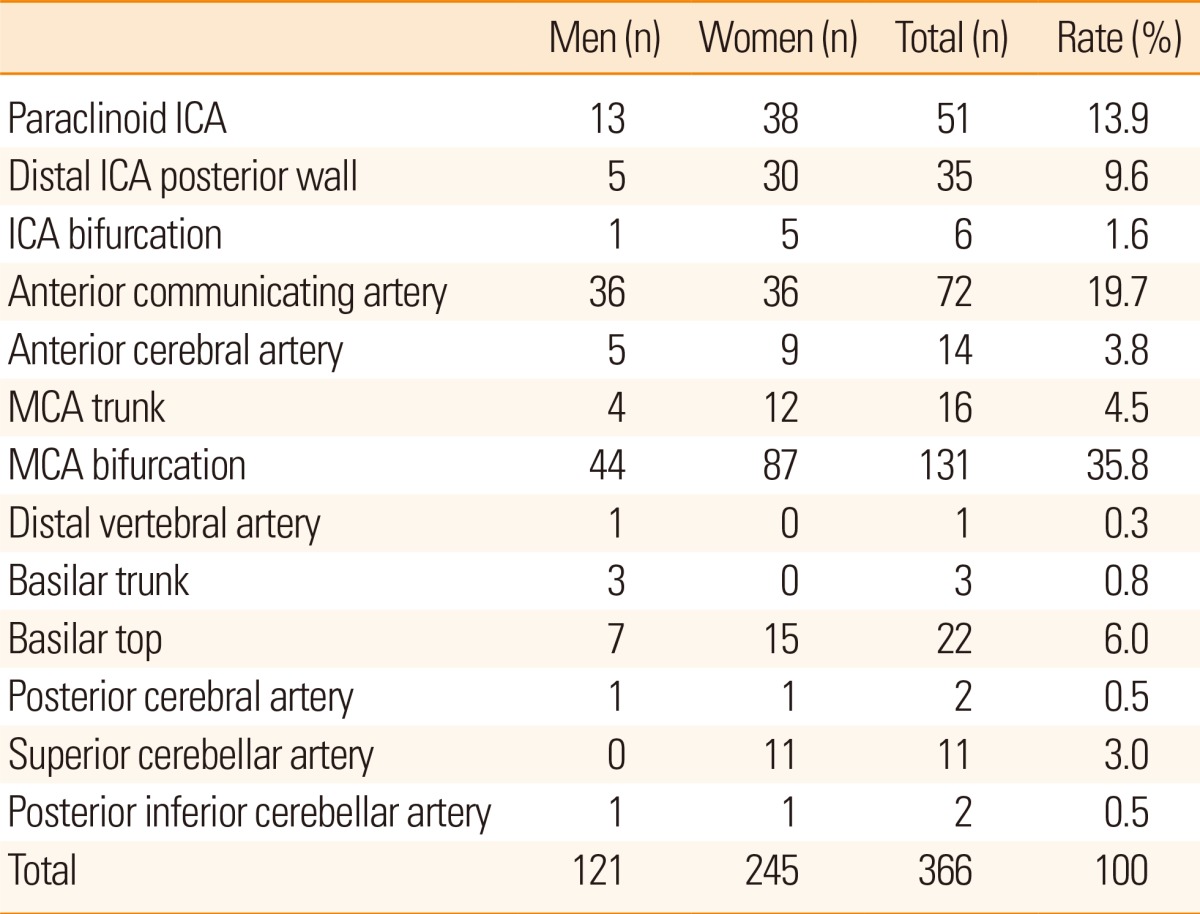
The most common aneurysm location was the bifurcation of the MCA (131/366, 35.8%) and the next most common location was the anterior communicating artery (72/366, 19.7%; Table 2). Other frequent sites were the paraclinoid ICA (51/366, 13.9%), distal ICA posterior wall (35/366, 9.6%), basilar top (22/366, 6.0%), and MCA trunk (16/366, 4.5%; Table 2). The bifurcation of the MCA was the most common location in both men and women (Table 2). There were 2 paraclinoid aneurysms in patients aged 20-29 years, and 2 MCA bifurcation aneurysms and 1 anterior communicating artery aneurysm in patients aged 30-39 years. In patients aged 40-49 years, the paraclinoid ICA was the most common site (n=10) followed by the anterior communicating artery (n=7), and bifurcation of the MCA (n =6). However, in patients aged 50-59 years, the MCA bifurcation was the most common site (n=23) followed by the anterior communicating artery (n=13) and paraclinoid ICA (n=10). A similar tendency was observed in patients aged 60-69 years (MCA bifurcation, 49; anterior communicating artery, 22; paraclinoid ICA, 15) and those aged 70-79 years (MCA bifurcation, 34; anterior communicating artery, 21; paraclinoid ICA, 8). Among patients aged ≥80 years, there were 3 MCA bifurcation aneurysms, 3 distal ICA posterior wall aneurysms, and 3 basilar top aneurysms.
False-positive cases
Ninety false-positive cases, in which the radiology report contained the term "aneurysm" in the initial search results but no aneurysm was found on re-interpretation of the MRA images, were recorded. This high rate of false positives was probably due to cases that had 'rule out aneurysm' noted in the radiological report being included in the initial search results. In some of the cases, it was difficult to determine whether the lesion was an aneurysm even after a third neuroradiologist was consulted, mainly because of image resolution or image artifacts. The most common sites for false positives were the anterior communicating artery (n=21) followed by the MCA bifurcation (n=20) and the posterior communicating artery (n=14). The causes of false positives were junctional dilatation (n=35), vascular ectasia (n=24), and broad-based focal bulging (n=14), which were all difficult to differentiate from MRA artifacts and vascular variations such as fenestration (n=17). The study design precluded us from detecting false-negative cases.
Follow-up results
The duration of clinical follow-up ranged from 94 days to 2,641 days, and the median follow-up duration was 1,256 days, which was shorter than we intended. This was due to our inability to strictly follow up these patients. During the follow-up period, 43 of the 330 patients (13.0%) were treated by clipping (n=32) or coiling (n=11). The treatment decision was made at various time points after the initial diagnosis of the aneurysm. Follow-up imaging was performed in 72 of the 287 untreated patients (25.1%; computed tomography angiography in 38, MRA in 29, and digital subtraction angiography in 5). Follow-up imaging showed enlargement of the aneurysm in 6 of the 72 patients (8.3%). Two of these were MCA bifurcation aneurysms, 2 were anterior communicating artery aneurysms, 1 was a distal ICA posterior wall aneurysm, and 1 was a basilar top aneurysm. Their initial size ranged from 4 to 6 mm.
Two patients presented with a subarachnoid hemorrhage during follow-up. One was a 61-year-old female who presented with a mild headache. The initial MRA showed a 9-mm aneurysm at the anterior cerebral artery A2-3 junction. Subsequent computed tomography angiography showed a broad-neck aneurysm with a height of 4 mm. The patient was reluctant to undergo surgery and was followed with imaging at regular intervals. Eight months after the initial diagnosis, the patient presented with a subarachnoid hemorrhage due to aneurysm rupture. The other patient was a 70-year-old female. The initial MRA was obtained to evaluate a brainstem infarction and showed a 3.5-mm left MCA bifurcation aneurysm. Twenty-seven months after her initial diagnosis, while on antiplatelet therapy, the patient presented with a subarachnoid hemorrhage due to aneurysm rupture.
Discussion
With the advent of less invasive vascular imaging techniques, the incidental discovery of asymptomatic intracranial aneurysms is increasing.8 The clinical consequence and symptomatic conversion rate of these aneurysms is unknown and is a pressing clinical issue.
There are many reports on the prevalence of unruptured saccular intracranial aneurysms.4,5,9,10 However, the prevalence varies significantly across reports, probably due to the diversity of study methods and/or ethnic differences among study populations. One angiography-based study in a large cohort of patients reported that the prevalence of asymptomatic intracranial aneurysms was 0.65%,9 and a recent meta-analysis reported a prevalence of 3.2%.4 A recent study evaluated the prevalence of unruptured intracranial aneurysms using 3-T MR imaging and reported a prevalence of 8.4%,6 which is substantially higher than previous reports.4,5 This can be explained by an increased rate of detection of aneurysms smaller than 3 mm due to the higher resolution of the 3-T MR system. We found a prevalence of 1.8%, which is somewhat lower than previous reports.4,10,11 There are several possible explanations for this difference. First, our study was not population-based and, although the patients were ethnically homogeneous, there may have been a bias in the study population. The lower rate might also be related to the low sensitivity of the 1.5-T MRA, a possibility supported by the small number of cases with aneurysms measuring less than 2 mm. Another important possible explanation is the existence of false-negative cases that were not included in the calculated prevalence. Because we identified incidental aneurysms based only on cases returned during our initial search of the radiology report database, there may have been a significant number of false-negative cases that were not identified. In light of these limitations, we urge caution in interpreting our MRA prevalence rate. However, our study represents the real-world experience of a single high-volume center with uniform non-invasive imaging that we believe can contribute to the overall view of prevalence, distribution of aneurysms in terms of sites, and age.
Our results confirmed a higher prevalence of intracranial aneurysms in women than in men.10 The prevalence of aneurysms in women increased with age, which is a well-known risk factor. One study hypothesized that this might be associated with the decrease in estrogen concentration and estrogen-receptor density that occurs in women during and after menopause.12 The prevalence of aneurysms in men older than 80 years was lower than that in men aged 70-79 years, although the difference was not statistically significant (P=0.30). Aneurysm prevalence was highest in women older than 80 years, and was higher in this group than in women aged 70-79 years. This is probably attributable to the relatively small sample of men aged over 80 years, which is due to the increased number of deaths in this age group.
A previous study demonstrated a gender difference in aneurysm location.13 However, there was no gender difference in aneurysm location in our study. Although some studies report that the paraclinoid ICA is the most common site for unruptured intracranial aneurysms,4,11,14 we found the most common site to be the MCA bifurcation, and this is consistent with other previous studies.15,16,17,18 Other frequent sites were the anterior communicating artery and the paraclinoid ICA. Before the age of 50, the most frequent aneurysm location was the paraclinoid ICA. However, the MCA bifurcation became the main predilection site after the age of 50. Significant increases in the prevalence of MCA bifurcation and anterior communicating artery aneurysms could be due to higher hemodynamic shear stress.19 Most aneurysms were between 2 mm and 7 mm in size. Aneurysms larger than 10 mm were rare, consistent with a previous report.9 Aneurysm enlargement on follow-up imaging was present in 6 cases, and aneurysm rupture during follow-up occurred in 2 cases. Enlargement or rupture therefore occurred in 2.8% of untreated patients. If all patients had been left untreated, the incidence of symptomatic conversion may have been higher than 2.8% as the decision to treat an aneurysm may have been based on the clinical perception of a high-risk lesion.
There were 90 false-positive cases that appeared in the initial database search. The causes of misclassification were junctional dilatation, vascular ectasia, broad-based focal bulging, and fenestration. False positives were most common in the anterior communicating artery. This is consistent with a recent report on the false-positive causes of aneurysms on MRA,20 and the causes of misclassification should be considered when analyzing MRA findings, especially for anterior communicating artery aneurysms. On the distal ICA, the differentiation of junctional dilatations from aneurysms may require additional image reconstruction. Many of the false positives in the current study could have been avoided with state-of-the-art MRA techniques, as many of the instances of vascular ectasia and bulging contours were a consequence of poor image quality.
Limitations inherent to the study design limit the conclusions that can be drawn from our results. The prevalence of incidental aneurysms may have been underestimated because we were unable to include false-negative cases, and the use of 1.5-T MRA data may have limited our ability to detect small aneurysms and thoroughly evaluate false-positive results due to limited resolution. In addition, the imaging follow-up data were limited because we could not strictly follow up these patients. We were therefore unable to obtain useful data on the natural history of incidental aneurysms.
Conclusions
We conducted a retrospective review of 18,237 patients who underwent MRA at our institution and found that the prevalence of unruptured intracranial aneurysms was 1.8%, which was significantly higher in women than in men. The prevalence increased with age in women, but not in men. Before the age of 50, the most frequent aneurysm location was the paraclinoid ICA. However, the MCA bifurcation became the main predilection site after the age of 50. Therefore, overall, the most common location for unruptured intracranial aneurysms was the bifurcation of the MCA. There were a number of false-positive cases that appeared in our initial search, and the most common location for false-positive cases was the anterior communicating artery.