Thrombolysis Improves Reperfusion and the Clinical Outcome in Tandem Occlusion Stroke Related to Cervical Dissection: TITAN and ETIS Pooled Analysis
Article information
Abstract
Background and Purpose
Despite the widespread adoption of mechanical thrombectomy (MT) for the treatment of large vessel occlusion stroke (LVOS) in the anterior circulation, the optimal strategy for the treatment tandem occlusion related to cervical internal carotid artery (ICA) dissection is still debated. This individual patient pooled analysis investigated the safety and efficacy of prior intravenous thrombolysis (IVT) in anterior circulation tandem occlusion related to cervical ICA dissection treated with MT.
Methods
We performed a retrospective analysis of two merged prospective multicenter international real-world observational registries: Endovascular Treatment in Ischemic Stroke (ETIS) and Thrombectomy In TANdem occlusions (TITAN) registries. Data from MT performed in the treatment of tandem LVOS related to cervical ICA dissection between January 2012 and December 2019 at 24 comprehensive stroke centers were analyzed. The primary endpoint was a favorable outcome defined as 90-day modified Rankin Scale (mRS) score of 0–2.
Results
The study included 144 patients with tandem occlusion LVOS due to cervical ICA dissection, of whom 94 (65.3%) received IVT before MT. Prior IVT was significantly associated with a better clinical outcome considering the mRS shift analysis (common odds ratio, 2.59; 95% confidence interval [CI], 1.35 to 4.93; P=0.004 for a 1-point improvement) and excellent outcome (90-day mRS 0–1) (adjusted odds ratio [aOR], 4.23; 95% CI, 1.60 to 11.18). IVT was also associated with a higher rate of intracranial successful reperfusion (83.0% vs. 64.0%; aOR, 2.70; 95% CI, 1.21 to 6.03) and a lower rate of symptomatic intracranial hemorrhage (4.3% vs. 14.8%; aOR, 0.21; 95% CI, 0.05 to 0.80).
Conclusions
Prior IVT before MT for the treatment of tandem occlusion related to cervical ICA dissection was safe and associated with an improved 90-day functional outcome.
Introduction
Since the adoption of mechanical thrombectomy (MT) for the treatment of patients with acute large vessel occlusion strokes (LVOS) [1], the role of prior intravenous thrombolysis (IVT) has been debated. Recent publications of the DIRECT-MT, SKIP, DEVT, and MR-CLEAN NO-IV trials and ongoing SWIFT-DIRECT trial are expected to bring part of the answer [2-5]. However, given the rarity of tandem occlusions in particular related to cervical internal carotid artery (ICA) dissection, these trials might not answer the question for this very specific subgroup. A high clot burden, the need for antiplatelet therapy if emergency ICA stenting is required, along with concerns about intracranial bleeding and extension or worsening of the arterial dissection are frequently raised arguments for avoiding prior IVT prior to MT. The efficacy and safety of IVT on ICA dissection-related strokes has been reported for all presentations, but without emphasis on tandem LVOS [6,7]. Tandem occlusion related to cervical ICA dissection is a singular entity with a specific therapeutic strategy regarding endovascular treatment and associated medical treatment [8-12]. This is an important question to address given the lack of data and various therapeutic alternatives based on local protocols. Therefore, this study assessed the impact of prior IVT on patients treated with MT for tandem LVOS related to cervical ICA dissection.
Methods
Study population
We performed a retrospective analysis of prospectively maintained multicenter observational registries. The study population was composed of two merged databases: Endovascular Treatment in Ischemic Stroke (ETIS; NCT03776877) from May 2012 to December 2019 and Thrombectomy In TANdem (TITAN) lesions from January 2012 to September 2016. ETIS is an ongoing prospective multicentric registry collecting data from consecutive patients benefiting from endovascular treatment for LVOS at six French comprehensive stroke centers [6]. The TITAN collaboration pooled individual data from non-randomized thrombectomy databases from 18 international centers for all consecutive anterior circulation tandem occlusions treated with MT [7]. Our analysis included patients with anterior circulation tandem occlusions due to cervical ICA dissection treated with an endovascular approach. Tandem occlusions were defined as cervical ICA occlusion or high-grade stenosis associated with a second distinct intracranial occlusion (intracranial segment of ICA, M1, or M2 segments of the middle cerebral artery [MCA]). ICA dissection diagnosis was based on angiographic features (per-thrombectomy angiographic aspect), imaging explorations demonstrating cervical ICA arterial wall hematoma, epidemiological data, and exclusion of differential stroke etiologies. Patients presenting with simultaneous multiple and confounding potential LVOS etiologies were excluded (for example, in case of co-existing atrial fibrillation).
Treatments
The indications for MT were based on the timeframes, imaging data, global comorbidities, and standard guidelines, after multidisciplinary discussion. IVT was performed within 4.5 hours after onset according to the international guidelines, using recombinant tissue plasminogen activator (alteplase, 0.9 mg/kg) in the absence of contraindications. MT was performed depending on the patient’s condition and local protocol. The endovascular strategy was left to the discretion of the interventionist, especially for intracranial MT approach (stent retriever alone, contact aspiration, or stent retriever+aspiration combination), emergency ICA stenting, and perioperative antiplatelet therapy. Depending on operator’s choice, peroperative aspirin intravenous bolus (250 to 500 mg) was administered. In case of cervical stenting, oral antiplatelet therapy was initiated after day-1 imaging according to the neurological condition, infarct extension, intracranial hemorrhage (ICH) and cervicocerebral arteries patency and chosen among the following possibilities: dual or simple antiplatelet therapy or none.
Data collection
The data used in this study are available from the corresponding author upon reasonable request. Clinical, imaging, timeline, and angiographic data were recorded. Trained research nurses assessed the modified Rankin Scale (mRS) score at 90 days, during face-to-face interviews or via telephone conversations with the patients, their relatives or their general practitioners. Imaging data were assessed by experienced (>10 years) senior investigators in participating centers. Favorable and excellent outcomes were respectively defined as a 90-day mRS score 0–2 and 0–1. Day-1 National Institutes of Health Stroke Scale (NIHSS) was recorded. Successful reperfusion was defined as modified Thrombolysis In Cerebral Infarction (mTICI) 2b-3. A first pass effect was defined as mTICI 2b-3 after one pass. Procedural complications (dissection, embolism in a new territory [ENT], and arterial perforation) and 90-day mortality were recorded. ICH was assessed according to the ECASS II classification. Symptomatic intracerebral hemorrhage (sICH) was defined as neurological deterioration (NIHSS worsening ≥4 points or death) along with ICH. Patency of the cervical carotid artery was evaluated on day-1 imaging follow-up (magnetic resonance angiography or computed tomography angiography).
Statistical analysis
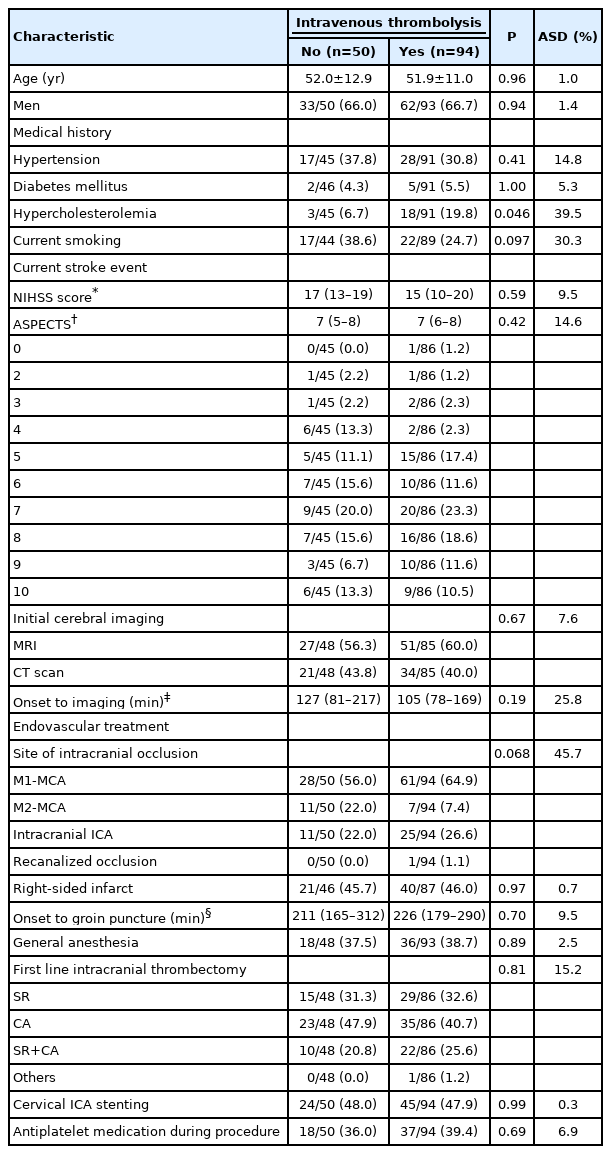
Categorical variables were expressed as frequencies and percentages. Quantitative variables were expressed as the mean±standard deviation or median (interquartile range) according to the normality of distribution which was assessed using histograms and the Shapiro-Wilk test. Patients were divided in two groups according to whether IVT was used before groin puncture. The patient and treatment characteristics were described and compared between the two study groups using chi-square test for categorical variables (or using Fisher’s exact test when expected cell frequency <5) and using Student's t-test for quantitative variables (or using Mann-Whitney U test for non-normal distribution); magnitudes of the betweengroup differences were assessed by calculating the absolute standardized differences; an absolute standardized difference <20% was interpreted as a small difference.
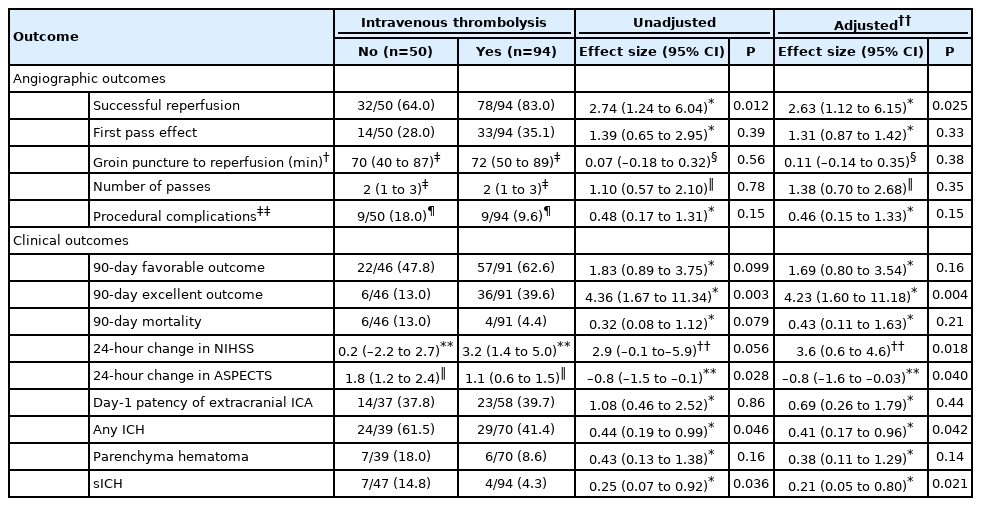
We compared outcomes in the two study groups before and after adjustment on onset to groin puncture, site of occlusion and cervical ICA stenting for angiographic outcomes and on pre-treatment NIHSS, pre-treatment Alberta Stroke Program Early CT Score (ASPECTS) and onset to groin puncture for clinical outcomes. A sensitivity analysis was done by excluding from the no-IVT group the patients outside the time window for IVT and those with extended infarct core on initial imaging. Binary outcomes (favorable and excellent outcomes, 90-day all-cause mortality, successful reperfusion, first pass effect, patency of the extracranial carotid artery, and procedural and hemorrhagic complications) were compared using logistic regression models (using penalized approach to account smaller number of events for procedural complication, all-cause mortality, parenchymal hematoma and symptomatic intracerebral hemorrhage); odds ratios (ORs) were calculated as the effect size using patients in the non-IVT group as the reference. The overall distribution of mRS and number of passes were compared using an ordinal logistic regression model (shift analysis mRS 5 and 6 and number of passes >5 pooled together); the common odds ratio (cOR) for a 1-point improvement in mRS and number of passes were derived from this model as the effects size. The groin-puncture-to-successful-reperfusion time and 24-hour NIHSS and ASPECTS changes were compared using a generalized linear regression model; mean betweengroup differences (IVT vs. non-IVT groups) were derived from this model as the effect size. The normality of model residuals was established after log-transformation of the groin puncture-to-successful-reperfusion time. Multivariate analyses were performed after handling missing values for confounding factors and outcomes using multiple imputation. The imputation procedure was performed with a regression-switching approach (chained equations with n=10) under the missing at random assumption using all baseline characteristics and study outcomes with a predictive mean matching method for continuous variables and logistic regression models (binary, ordinal, or multinomial) for categorical variables. Estimates obtained with the different imputed datasets were combined using Rubin’s rules.
Statistical testing was done at the two-tailed α level of 0.05. The data were analyzed using SAS release 9.4 (SAS Institute, Cary, NC, USA).
Ethics statement
Ethics approval was obtained from the local institutional review board. Informed consent was obtained for all the patients.
Results
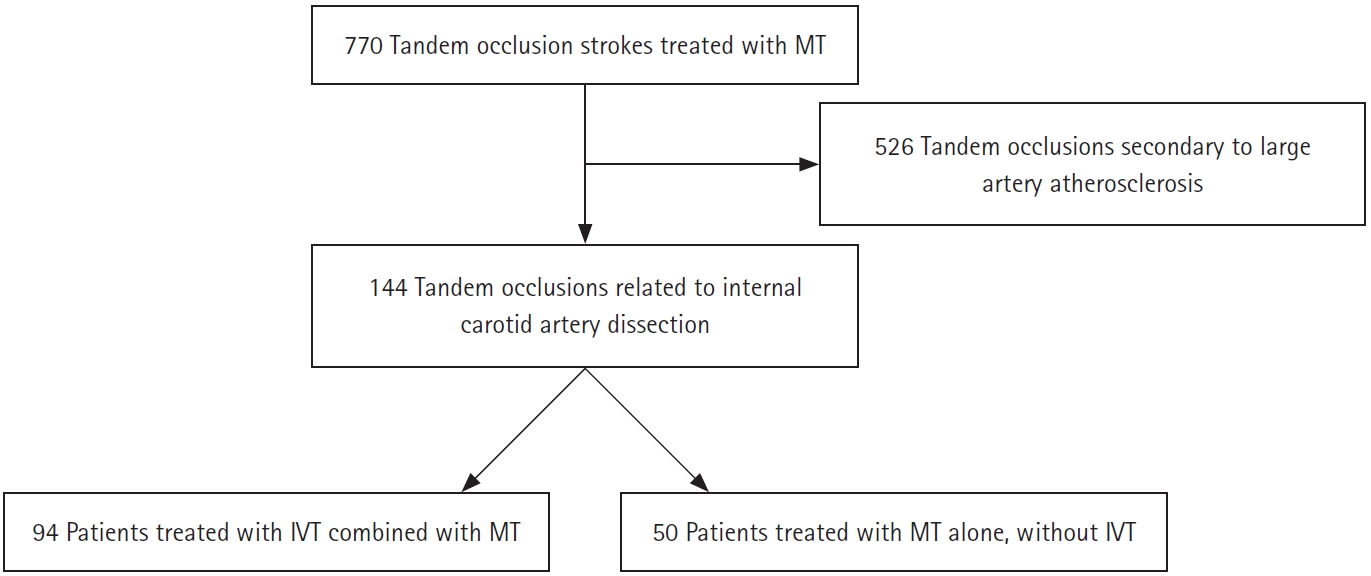
Of the 770 LVOS patients with tandem lesions enrolled in the TITAN (n=369) and ETIS (n=401) registries, this study included 144 patients with cervical ICA dissection, of whom 94 (65.3%) underwent IVT (Figure 1). Table 1 summarizes the main patient and treatment characteristics according to IVT. The two groups were well balanced, except for hypercholesterolemia, current smoking, and site of occlusion (M2-MCA occlusion less frequent in IVT group). The reasons for no-IVT were outside the time window (n=15), unknown onset (n=7), extended infarction (n=8), interdisciplinary choice (n=12), prior double antiplatelet therapy (n=1), international normalized ratio >1.7 (n=1), recent abdominal surgery (n=1), and missing data (n=5).
As shown in Table 2, a higher rate of favorable outcome (pre-specified as primary outcome) was observed in the IVT group (62.6% vs. 47.8%) although the difference did not reach statistical significance (OR, 1.83; 95% confidence interval [CI], 0.89 to 3.75). When the overall degree of disability was analyzed, the difference in favor of the IVT group reached significance (Figure 2), and also when an excellent outcome was considered (OR, 4.36; 95% CI, 1.67 to 11.34). Similar results were found after pre-specified adjustment for pre-treatment NIHSS, ASPECTS, and onset to groin puncture time, with an adjusted odds ratio (aOR) for the IVT group of 2.44 (95% CI, 1.28 to 4.61) for a 1-point improvement in the mRS score and 4.23 (95% CI, 1.60 to 11.18) for an excellent outcome. The rate of successful reperfusion was higher in the IVT group (83.0% vs. 64.0%; aOR, 2.63; 95% CI, 1.12 to 6.15). Regarding other clinical efficacy outcomes, patients in the IVT group had a significantly greater reduction in day-1 NIHSS (adjusted mean difference, 3.6 points; 95% CI, 0.6 to 4.6) and a lower worsening in day-1 ASPECTS (adjusted mean difference, –0.8 points; 95% CI, –1.6 to 0.03). sICH and any ICH were also significantly less frequent in IVT group, with a respective aOR of 0.21 (95% CI, 0.05 to 0.80) and 0.41 (95% CI, 0.17 to 0.96). Similar non-significant differences in all-cause mortality and parenchymal hematoma rates were found. Peroperative complication rates were comparable between groups (18.0% vs. 9.6%; aOR, 0.46; 95% CI, 0.15 to 1.33). Detailed complications were as follow: nine cases in the no-IVT group (eight ENT and one perforation) and nine in the IVT group (three ENT, five perforations, and one extension and worsening of ICA dissection).
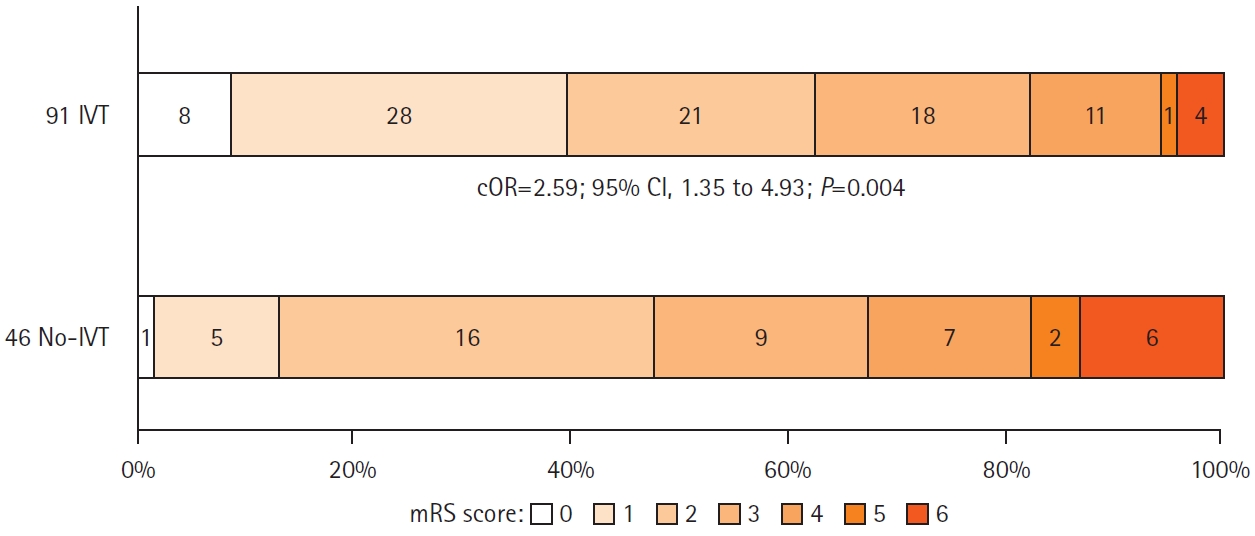
Distribution of 90-day modified Rankin Scale (mRS) scores according to use or not of intravenous thrombolysis (IVT) prior endovascular treatment. Common odds ratio (cOR) indicates for 1-point improvement in 90-day mRS score (after combining 5 and 6 together). CI, confidence interval.
The results of sensitivity analysis, after exclusion from the no-IVT group of 15 patients outside the IVT time window and eight with large infarction on initial imaging, are presented in Supplementary Table 1. A significantly higher rate of successful reperfusion was observed in the IVT group (83.0% vs. 63.0%; unadjusted OR, 2.87; 95% CI, 1.11 to 7.41). Notably, ICH, parenchymal hematoma, and sICH rates were comparable between groups with respective unadjusted ORs of 0.86 (95% CI, 0.31 to 2.36), 0.50 (95% CI, 0.12 to 2.02), and 0.78 (95% CI, 0.11 to 5.40).
Discussion
Two important findings emerged from this individual patient pooled study. First, bridging therapy resulted in a better functional outcome compared to MT alone. Second, the use of IVT before MT was associated with lower odds of ICH and sICH.
The recent publication of the DIRECT-MT, SKIP, and DEVT trials and the presentation of MR-CLEAN NO-IV trial results revealed evidence in favor of the non-inferiority of MT alone versus bridging therapy in LVOS [2-5]. The benefit of systematic use of IVT prior to MT remains debated and more evidence is needed before current guidelines are modified [8]. In addition, rare and specific occlusion subtypes, such as tandem occlusions, are either underrepresented or often excluded [2] in such trials, emphasizing the importance of results from large retrospective cohorts.
In our population, successful reperfusion was more frequent after bridging therapy. This clinical benefit of IVT has been previously reported [13,14], but was not observed in the recent trials [2-5]. Still, especially considering drip-and-ship admissions, IVT is associated with an improved rate of intracranial recanalization before MT, positively influencing clinical outcome [15]. Interestingly, in our study, one case of early recanalization after IVT was observed whereas no case was noted in the no-IVT group. In our study, for tandem LVOS related to cervical ICA dissection, bridging therapy seems to improve functional outcome compared to MT alone.
ICH is a feared complication of IVT. In the treatment of tandem occlusion, combining IVT with antiplatelet therapy, especially in cases of acute stenting, is a questionable approach due to the risk of hemorrhagic complications. Here, the rates of stenting and antiplatelet agent use were similar between groups and there was no increased risk of ICH in the IVT group. Conversely, sICH and any ICH were more frequent in the no-IVT group. This finding might be related to a higher proportion of patient outside the time window for IVT and/or with larger initial infarct core on initial imaging in the no-IVT group. This hypothesis is confirmed the sensitivity analysis after exclusion of these patients demonstrating no difference in ICH and sICH risk between IVT and no-IVT groups. The worse recanalization and higher infarct expansion rates in the no-IVT group may also explain an increased ICH risk. Indeed, lower recanalization rates and poorer day-1 ASPECTS have previously been proven to be predictive of higher hemorrhagic risk [6]. Also, even if there role can be evocated, antiplatelet medications were reported safe in the treatment of tandem occlusions, regardless of IVT status [16].
One concern with IVT for such indications is the fear of extension and worsening of the dissection [17,18]. This is supported by the possible lysis of the arterial wall hematoma allowing progression of the dissection and potential ischemic worsening. We did not find any evidence supporting this phenomenon or its consequences. In particular, the day-1 patency of the cervical ICA was comparable between groups.
Our study has several limitations mostly related to its retro-spective design. The endovascular strategies, stenting protocol, and perioperative antiplatelet regimens were not standardized. Decision to administer IVT was left to the discretion of local teams. The study was conducted over an extended timespan which can constitute a source of bias regarding potential evolution of therapeutic strategies. The absence of core lab review of imaging and centralized measurements of infarct core, penumbra, and mismatch ratio might also be a limitation. Due to the rarity of tandem occlusion related to dissection and the difficulty in forming large subgroups, the statistical analysis may lack power without no formal sample calculation. Due to exploratory nature of the present study, comparisons in outcomes were not adjusted for multiplicity and we therefore could not excluded false positive findings. For all these reasons, the present results should be interpreted only as hypothesis generating and should be replicated in further larger studies.
Conclusions
IVT before MT for tandem occlusions related to cervical ICA dissection seems to be safe and associated with a better functional outcome. In the absence of contraindication and within the recommended time window, prior IVT may still be considered as part of the acute reperfusion therapy for tandem occlusions related to cervical ICA dissection.
Supplementary materials
Supplementary materials related to this article can be found online at https://doi.org/10.5853/jos.2020.04889.
Outcomes according to intravenous thrombolysis prior endovascular treatment after exclusion in the no-IVT group of 15 patients outside the time window for IVT and eight with large ischemic core on initial imaging
Notes
Disclosure
The authors have no financial conflicts of interest.
ETIS investigators
Michel Piotin, Raphaël Blanc, Simon Escalard, Jean-Philippe Desilles, Benjamin Maïer, Gabriele Ciccio, Stanislas Smajda, Mikael Mazighi, François Delvoye, Robert Fahed, Mikael Obadia, Candice Sabben, Ovide Corabianu, Thomas de Broucker, Didier Smadja, Sonia Alamowitch, Olivier Ille, Eric Manchon, PierreYves Garcia, Guillaume Taylor, Malek Ben Maacha.
Frédéric Bourdain, Jean-Pierre Decroix, Adrien Wang, Serge Evrard, Maya.Tchikviladze, Bertrand Lapergue, Oguzhan Coskun, Arturo Consoli, Federico Di Maria, Georges Rodesh, Morgan Leguen, Marie Tisserand, Fernando Pico, Haja Rakotoharinandrasana, Philippe Tassan, Roxanna Poll.
Gaultier Marnat, Florent Gariel, Xavier Barreau, Jérôme Berge, Patrice Menegon, Igor Sibon, Ludovic Lucas, Stéphane Olindo, Pauline Renou, Sharmila Sagnier, Mathilde Poli, Sabrina Debruxelles, François Rouanet, Thomas Tourdias, Jean-Sebastien Liegey, Isabelle Quintana, Tristan Kerdraon.
Romain Bourcier, Lili Detraz, Benjamin Daumas-Duport, Pierre-Louis Alexandre, Monica Roy, Cédric Lenoble, Vincent L’allinec, Jean-Baptiste Girot, Hubert Desal.
Benjamin Gory, Fatiha Bechiri, Sarah Guy, Serge Bracard, René Anxionnat, Marc Braun, Anne-Laure Derelle, Romain Tonnelet, Liang Liao, François Zhu, Emmanuelle Schmitt, Sophie Planel, Sébastien Richard, Lisa Humbertjean, Gioia Mione, Jean-Christophe Lacour, Gabriela Hossu, Marine Beaumont, Mitchelle Bailang, Gérard Audibert, Marie Reitter, Agnès Masson, Lionel Alb, Adriana Tabarna, Marcela Voicu, Iona Podar, Madalina Brezeanu.
Vincent Costalat, Cyril Dargazanli, Federico Cagnazzo, Alain Bonafé, Grégory Gascou, Pierre-Henri Lefèvre, Imad Derraz, Carlos Riquelme, Caroline Arquizan, Nicolas Gaillard, Isabelle Mourand, Lucas Corti.
TITAN investigators
Mohammad Anadani, Alejandro M. Spiotta, Ali Alawieh, Francis Turjman, Michel Piotin, Diogo C. Haussen, Raul G. Nogueira, Panagiotis Papanagiotou, Adnan H. Siddiqui, Bertrand Lapergue, Franziska Dorn, Christophe Cognard, Marc Ribo, Marios N. Psychogios, Marc Antoine Labeyrie, Mikael Mazighi, Alessandra Biondi, René Anxionnat, Serge Bracard, Sébastien Richard, Benjamin Gory, Jonathan Andrew Grossberg, Adrien Guenego, Julien Darcourt, Isabelle Vukasinovic, Elisa Pomero, Jason Davies, Leonardo Renieri, Corentin Hecker, Maria Muchada Muchada, Arturo Consoli, Georges Rodesch, Emmanuel Houdart, Raymond Turner, Aquilla Turk, Imran Chaudry, Johanna Lockau, Andreas Kastrup, Raphaël Blanc, Hocine Redjem, Daniel Behme, Hussain Shallwani, Maurer Christopher, Gioia Mione, Lisa Humbertjean, Jean-Christophe Lacour, François Zhu, AnneLaure Derelle, Romain Tonnelet, Liang Liao.
Acknowledgements
On behalf of the Endovascular Treatment in Ischemic Stroke (ETIS) and Thrombectomy In TANdem (TITAN) Investigators