Smoking is Not a Good Prognostic Factor following First-Ever Acute Ischemic Stroke
Article information
Abstract
Background and Purpose
There is evidence that smoking increases stroke risk; however, the effect of smoking on functional outcome after stroke is unclear. The aim of this study was to explore the effect of smoking status on outcome following acute ischemic stroke.
Methods
We assessed 1,117 patients with first-ever acute cerebral infarction and no prestroke disability whose functional outcome was measured after three months. A poor outcome was defined as a modified Rankin Scale score of ≥2. Smoking within one month prior to admission was defined as current smoking. Our analysis included demographics, vascular risk factors, initial National Institutes of Health Stroke Scale (NIHSS) score, stroke subtype, onset-to-admission time, thrombolytic therapy, initial blood pressure, and prognostic blood parameters as covariates.
Results
At baseline, current smokers were predominantly male, approximately 10 years younger than non-smokers (mean age, 58.6 vs. 68.3 years), and less likely to have hypertension and atrial fibrillation (53.9% vs. 65.4% and 8.7% vs. 25.9%, respectively), with a lower mean NIHSS score (4.6 vs. 5.7). The univariate analyses revealed that current smokers had a better functional outcome and significantly fewer deaths at three months follow-up when compared with non-smokers (functional outcome: 64.0% vs. 58.4%, P=0.082; deaths: 3.0% vs. 8.4%, P=0.001); however, these effects disappeared after adjusting for covariates (P=0.168 and P=0.627, respectively).
Conclusions
In this study, smoking was not associated with a good functional outcome, which does not support the paradoxical benefit of smoking on functional outcome following acute ischemic stroke.
Introduction
Smoking is a modifiable and established risk factor for stroke.1,2 The age-standardized prevalence of current smoking in Korean adults is substantially high when compared with the US population, in particular in men (48.1% of Korean men vs. 21.2% of US men).3,4 The population attributable risk of smoking for stroke is also high in Korean men (26.5%).5 Therefore, antismoking campaigns are an important health-related intervention for stroke prevention in Korea.
Despite the increased risk of stroke from smoking, there is debate regarding its effect on functional outcome following acute ischemic stroke. Current smoking prior to stroke is a poor6,7,8 or neutral prognostic factor according to the results of some studies.9,10,11,12,13 Surprisingly, smoking has been reported as a beneficial prognostic factor in acute ischemic stroke.14,15,16,17 It is postulated that chronic exposure to smoking or nicotine may promote angiogenesis or ischemic preconditioning with enhanced collateral flows. In addition, there might be a greater susceptibility to spontaneous or therapeutic thrombolysis of cerebral thrombi in smokers.
Due to these conflicting results, we aimed to explore whether cigarette smoking was independently associated with the functional outcome of patients after acute ischemic stroke using our prospective registry database. In addition, we investigated whether any smoking effect differed across subgroups, including ischemic stroke subtypes.
Methods
Study design and subjects
Subjects were consecutive patients with first-ever ischemic stroke and without previous functional disability (modified Rankin Scale [mRS] score >1) who were admitted to Hallym University Medical Center within 7 days of symptom onset between October 2007 and July 2012. The data were populated from the Hallym Stroke Registry, a prospective hospital-based stroke database. The Institutional Review Board of Hallym University Medical Center approved the stroke registry and waiver of informed consent from registered patients. Patient information was anonymized and de-identified prior to analysis. Of the 1,248 patients who met our inclusion criteria, we excluded 131 patients (10.5%) because they did not have 3-month functional outcome data. Therefore, 1,117 subjects were included in the outcome analysis.
Smoking status and covariate factors
We defined current smokers as those with any history of smoking within one month prior to index stroke. We subdivided non-smokers into former-smokers and never-smokers. Former-smokers did not smoke at least one month before the index stroke and never-smokers were those with no smoking history.
We included the following covariates in our analysis: demographics, body mass index (BMI), vascular risk factors, initial National Institutes of Health Stroke Scale (NIHSS) score, stroke subtype (according to Trial of ORG 10172 in Acute Stroke Treatment [TOAST] criteria), onset-to-admission time, tissue plasminogen activator (tPA) therapy, mean value of initial systolic blood pressure (systolic BP), and prognostic blood parameters including blood glucose, low density lipoprotein (LDL) cholesterol, white blood cell (WBC) count, and hemoglobin count. Blood glucose and LDL cholesterol were measured in a fasting state during the first morning after admission.
Hypertension was assigned based on the presence of one or more of the following: history of hypertension and antihypertensive medication, repeated BP measurements >140/90 mmHg at 1 week intervals, or evidence of hypertensive damage on echocardiogram. Diabetes was assigned if there was a history of diabetes mellitus while also receiving anti-hyperglycemic therapy, fasting blood glucose level >126 mg/dL, or postprandial blood glucose level >200 mg/dL. Hyperlipidemia was assigned if there was a history of hyperlipidemia with concurrent pharmacotherapy or fasting LDL cholesterol level >160 mg/dL (or total cholesterol >240 mg/dL if LDL was not available).
Outcome assessment
Functional outcome and death were assessed at three months after stroke through face-to-face or telephone interview with patients or caregivers. Functional outcome was measured by mRS. A poor functional outcome was defined as an mRS score ≥2.
Statistical analysis
We compared the baseline characteristics of current smokers with non-smokers, former-smokers, and never-smokers (Table 1). We also compared the baseline characteristics of patients with a 3 month post-stroke mRS score available and those lost to follow-up (Supplementary Table 1). The χ2 test and t test were used for categorical and continuous variables, respectively. NIHSS scores were split into quartiles. We divided BMI into high (≥25 kg/m2), normal (<25 but >20 kg/m2), and low BMI (≤20 kg/m2).
Univariate and binary logistic regression analyses were used to determine the association between smoking status and functional outcome at three months. All variables in the univariate analysis were included in the multivariate analysis because they were potential prognostic factors. Small vessel disease was regarded as the reference among stroke subtypes. Those with undetermined and other determined stroke subtypes were not included in the univariate and multivariate analyses.
We also evaluated the effect of smoking through subgroup analysis of sex, age, stroke subtype, risk factors (hypertension, diabetes mellitus, hyperlipidemia, and atrial fibrillation), BMI, and tPA therapy.
We excluded the cases with missing data from all analyses. The significance level was set at P<0.05 for all statistical analyses.
Results
Baseline characteristics
The 1,117 patients included 639 men (57.2%) and 487 women with a mean age of 65.3 (±13.5) years. Median NIHSS was 3 (interquartile range 1-7). Mean onset-to-admission time was 28.6 (±37.1) hours. Data for atrial fibrillation were missing in 3 (0.3%), smoking status in 4 (0.36%), BMI in 33 (3.0%), WBC in 1 (0.1%), LDL-C in 22 (2.0%), and fasting glucose in 98 (8.8%) patients.
The study participants included 336 current smokers (30.2%), 126 former-smokers (11.3%), and 651 never-smokers (58.5%). Each group had significantly different baseline characteristics. Current smokers were mostly male and younger than former-smokers or never-smokers. Atrial fibrillation and cardioembolic stroke were significantly less frequent in current smokers than in former-smokers or never-smokers. Detailed comparisons of the baseline characteristics of the groups are presented in Table 1.
Smoking status was not significantly different between end-point patients and those lost to follow-up. However, lost to follow-up patients had more severe strokes (higher mean initial NIHSS score) and had longer onset-to-admission times (Supplementary Table 1).
Smoking status and initial stroke severity
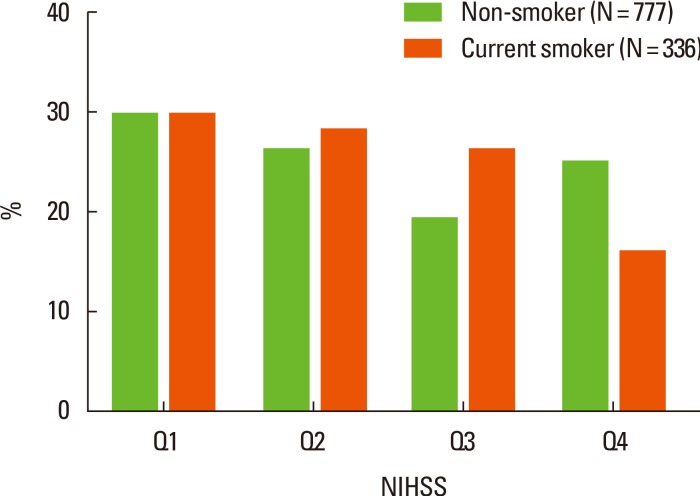
Initial mean NIHSS score was lower in current smokers than in former-smokers or never-smokers (Table 1). Figure 1 shows the frequency distribution of the quartiles of NIHSS scores in current smokers and non-smokers. Interestingly, the multivariate analysis revealed that current smoking was an independent predictor of severe stroke (defined as an NIHSS score of ≥4, Table 2). When we defined severe stroke as an NIHSS score of ≥6, current smoking remained as a significant predictor of severe stroke (Supplementary Table 2).
Smoking status and functional outcomes
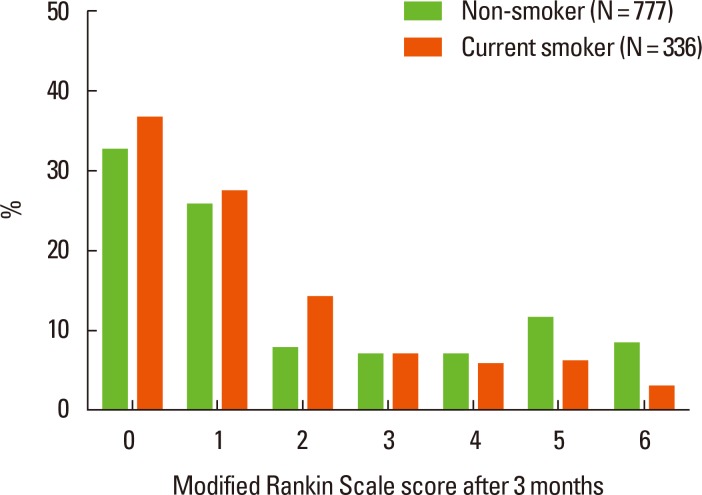
The univariate analysis showed that current smoking status was more frequent among patients with a good functional outcome than in those with a poor outcome (32.1% vs. 27.3%, P=0.082). A good functional outcome was more common in current smokers than non-smokers: 64.0% (215/336) of current smokers and 58.4% (454/777) of non-smokers (57.9% [73/126] of former-smokers and 58.5% [381/651] of never-smokers) had a good functional outcome. However, these differences were not statistically significant. Figure 2 shows the frequency distribution of the mRS scores in current smokers and non-smokers.
The percentage of smokers was lower among the patients who died within three months compared with that in the survivor group (13.3% vs. 31.4%, P=0.001). In particular, 3-month mortality was higher in non-smokers than in current smokers: 8.4% (65/777) vs. 3.0% (10/336).
Other parameters, including age, sex, BMI, vascular risk factors, initial NIHSS score, stroke subtype, onset-to-admission time, tPA therapy, mean value of initial systolic BP, and blood parameters were primarily associated with patients' outcomes. The results of the univariate analysis are described in Supplementary Tables 3 and 4.
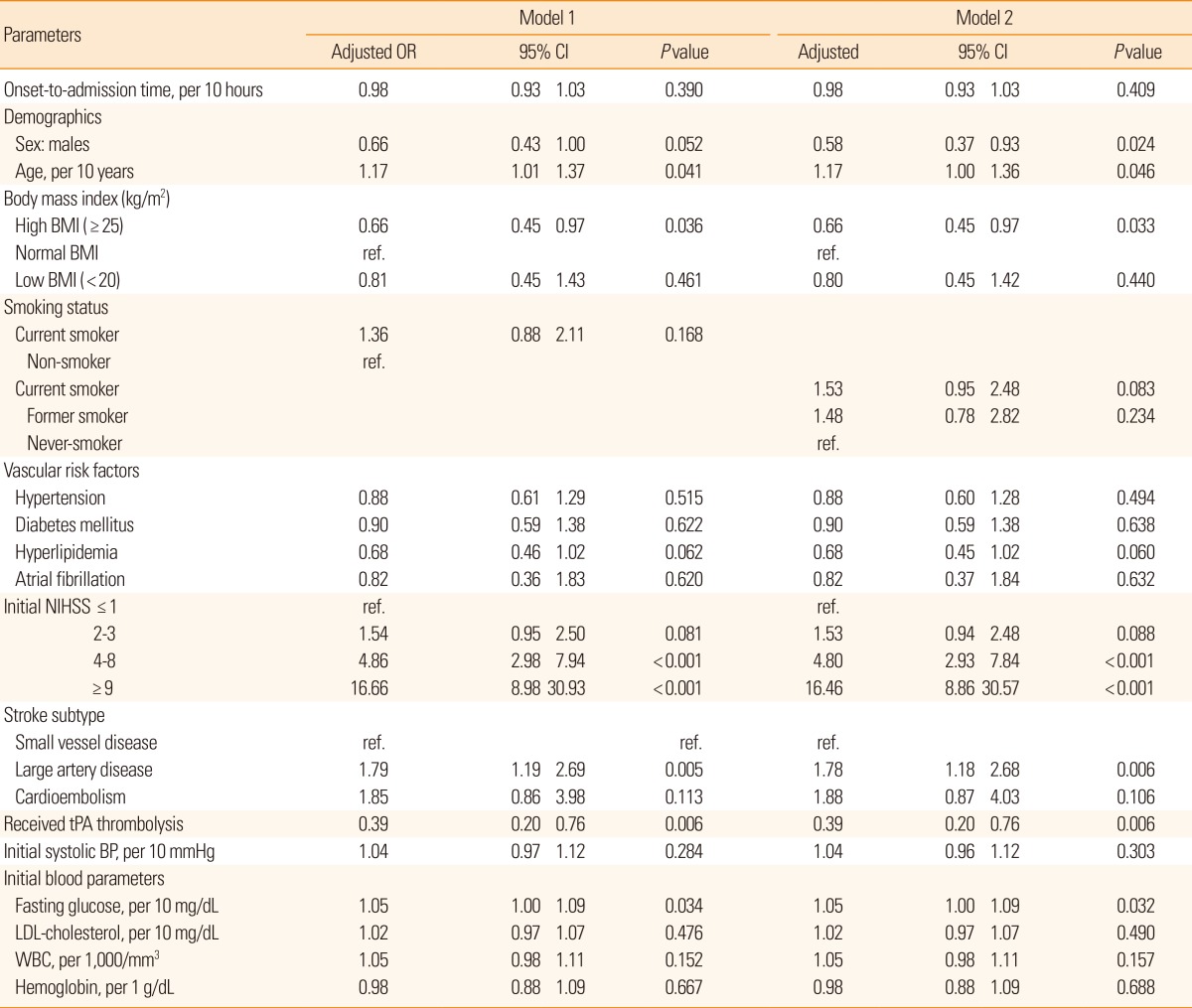
In the multivariate analysis, smoking status was not associated with functional outcome and death. Age, initial high stroke severity, large artery disease, high fasting blood glucose, and WBC count were independent prognostic factors for a poor outcome. Male gender, high BMI, and tPA therapy were good prognostic factors (Tables 3 and 4).
Effect of smoking status in the subgroups
In most subgroups, the functional outcome was not significantly different between current smokers and non-smokers (Figure 3 and Supplementary Table 5); however, current smoking was associated with a poor functional outcome in patients treated with tPA therapy and with a low BMI (P=0.036 and P=0.031, respectively; Supplemental Table 5).

The effect of current smoking on functional outcome in different subgroups (current smoker versus non-smoker). The diamond represents the point estimate of the adjusted odds ratio and the horizontal line represents the 95 percent confidence intervals. Poor functional outcome was defined as modified Rankin Scale score of ≥2, measured at three months after stroke. All variables in the main analysis were included as confounders in this subgroup analysis.
Discussion
The baseline characteristics of patients experiencing acute ischemic stroke were significantly different according to smoking status. Current smokers were predominantly young and male with a relatively benign baseline. A history of hypertension and atrial fibrillation were less frequent when compared with non-smokers. In addition, initial stroke severity was milder in current smokers. However, after adjustment for confounding variables, current smoking was independently associated with severe stroke. In the univariate analyses revealed that smokers displayed a good functional outcome and significantly less mortality within three months when compared with non-smokers; however, smoking status was not independently related with functional outcome after adjustment for demographics and other prognostic indicators. These results were consistent in subgroups that were stratified by gender and stroke subtype.
The baseline profile of smokers in our study corresponded well to the results from previous studies.6,8,9,10,12,13,14,17 The younger mean age in smokers may be associated with smokers having strokes at an earlier age than non-smokers. Cigarette smoking is known to be an independent risk factor for atrial fibrillation and hypertension.18,19 However, as with previous studies,6,8,9,10,12,14,17 our research reported a lower prevalence of atrial fibrillation and hypertension in current smokers with acute ischemic stroke when compared with non-smokers. Our univariate analysis showed a milder stroke severity in current smokers compared with non-smokers. This might be attributed to the lower frequency of atrial fibrillation (thereby low prevalence of cardioembolic stroke) in current smokers than that in non-smokers. These counterintuitive findings can be partially explained by a selective survival bias and demographic differences; atrial fibrillation and hypertension may be less frequent in current smokers because they are approximately 10 years younger than non-smokers in our study. In addition, patients with vascular risk factors may be less inclined to smoke as they have encountered non-smoking education or counseling more often. Other unmeasured confounding factors may be involved in different baseline characteristics that are related to smoking status. Other unidentified health problems may exist that are underlying drivers of non-smoking behavior. It is a more reasonable hypothesis that health-related behavior modification or health-seeking behavior is driving these findings rather than the presence of a protective effect of smoking.
The variables that are known to be potential prognostic factors required careful statistical analysis as there was a wide discrepancy between the baseline characteristics of the different groups. In both our study and the literature, there are some important limitations in performing this task; despite including a considerable number of well-known prognostic factors in the multivariable analysis, unmeasured confounding variables may contribute to erroneous conclusions. We believe that the utility of using smoking as a prognostic tool is controversial as it may be largely attributed to unaccounted confounding variables.
Socioeconomic status is likely to be an important confounder that has not been measured in this study. Low socioeconomic status may be a predictor of poor outcome after stroke.20 Most previous studies have not included socioeconomic status in multivariable analyses.7,8,9,10,11,12,13,14,15,16,17 Kim et al.6 included socioeconomic status when analyzing the effect of baseline smoking status on the long-term risk of a poor outcome (first vascular event or death) in stroke patients. Although the univariate analysis showed that smokers have a better outcome than people who have never smoked (P=0.03), they had a greater risk of a poor outcome in the multivariate analysis (P=0.003). In addition, changes in smoking habits after the index stroke may affect stroke outcome. These were not evaluated in our research or previous studies.6,7,8,9,10,11,12,13,15,16,17 Although little is known about the effects of smoking cessation on short-term stroke recovery, smoking cessation as a long-term protective measure with respect to mortality and cardiovascular events is well defined.21,22 Current smokers with mild or no disability may continue smoking compared with those who experienced severe stroke,23 which may affect the functional outcome. Moreover, smoking habits can be affected by previous stroke or functional status, which may be an important confounding factor.15,23 In previous studies, smokers are less likely to report a history of prior strokes than non-smokers (20.6% vs. 33.8%);15 therefore, we only included patients with first-ever stroke and no functional disability to exclude their confounding effects. However, many previous studies did not exclude or adjust for these factors in the multivariate analysis.7,8,10,12,13,16,17
Two previous studies have shown that patients with thrombolytic therapy experience a paradoxical short-term benefit of smoking.16,17 Smoking was associated with either improved recanalization16 or short-term improvement of stroke severity.17 The authors of these studies have hypothesized that smokers may have thrombi with more dissoluble characteristics. Nevertheless, smoking status was not associated with the functional outcome at three months. No association between baseline smoking status and functional outcome is reproduced in other studies.9,12 In these studies, despite an association with good outcomes in descriptive or univariate analyses, current smoking was not independently associated with the functional outcome after adjusting for confounding variables. In our subgroup analysis, smokers with thrombolytic therapy tended to have a poorer outcome than non-smokers; however, the false positive rates associated with the application of multiple statistical tests may be high; the probability of one or more false positive findings when 9 subgroup tests are performed is about 40 percent.24 Despite this limitation, our finding suggested that smoking might not predict improved functional outcome after thrombolytic therapy, irrespective of a reported high recanalization rate.16
A previous study has discussed the differing prognostic effect of smoking in relation to stroke subtypes: smoking is found to be beneficial only in atherosclerotic stroke.15 In contrast, another study has shown that male smokers with acute atherosclerotic stroke have an increased risk of a poor 90-day functional outcome, independent of other variables.7 Smoking was not independently associated with the functional outcome in atherosclerotic stroke in our subgroup analysis. The effect of smoking on functional outcome according to stroke subtypes needs to be further studied.
Our study has limitations. First, there may be a selection bias as patients lost to follow-up were excluded. These patients may have a poor functional outcome; in our data, this group had greater initial stroke severity and higher glucose levels (Supplementary Table 1), which are well known poor prognostic factors. Nevertheless, smoking status between the included and excluded (patients lost to follow-up) patients were not significantly different in our study (Supplementary Table 1); therefore, excluding these data would not significantly affect our study findings. Second, similar to many previous studies, it was not possible to analyze the dose effect of smoking because detailed information about quantity and potency was not available. Moreover, given that the risk of cardiovascular events increases with the number of cigarettes smoked, the impact of smoking on stroke outcome is likely to differ between light and heavy smokers.1,2,25 This effect needs to be explored in more detail. Third, there were some missing data in our study, in particular the fasting blood glucose levels and body mass index. These omissions were random because most baseline characteristics, such as smoking status and initial stroke severity were not different relative to the presence of blood parameters (Supplementary Table 6). Fourth, although we included only patients without prior stroke or functional disability, smokers are likely to have other medical problems, such as coronary artery and pulmonary disease. These medical problems may contribute to the functional outcome after stroke. Unfortunately, we were not able to adjust for these potential confounding factors.
Conclusions
In this study, smoking was not associated with a good functional outcome. This does not support the paradoxical benefit of smoking on functional outcome following acute ischemic stroke, which might be attributed to unmeasured confounding variables.
Acknowledgements
We thank Dr. Sean HaShemi (Department of Otolaryngology, Head and Neck Surgery, Boston University Medical Center, 820 Harrison Blvd, FGH 4th Floor, Boston, MA USA 02118, email: Sean.Hashemi@bu.edu) for helping us prepare this manuscript.
Notes
This study was supported by a grant from the Korea Health 21 R&D Project, Ministry of Health, Welfare and Family Affairs, Republic of Korea (HI10C2020).
The authors have no financial conflicts of interest.
References
Supplementary Material
Supplementary Table 1
Baseline characteristics of patients lost to follow-up
Supplementary Table 2
Factors associated with severe stroke (≥ NIHSS 6) in the multivariate analysis
Supplementary Table 3
Factors associated with functional outcomes in the univariate analysis
Supplementary Table 4
Factors associated with 3-month death in the univariate analysis
Supplementary Table 5
Adjusted odd ratio for poor functional outcome in different subgroups
Supplementary Table 6
Baseline characteristics of patients with missing data